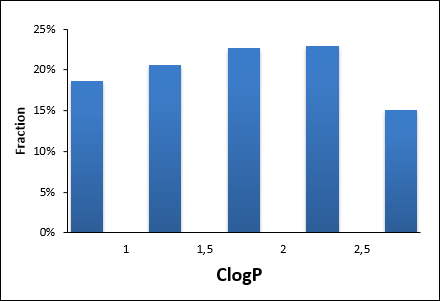
Enantiomeric pairs of covalent electrophilic fragments
960 compounds
Covalent chemical probes have become a valuable tool in drug discovery within the last few years. New technologies and development of fast mass spectrometric screening and imaging open a new horizon for proteome-wide screening and proteomics discoveries. The impressive number of successful applications brought aspiration to the discovery and synthesis of new covalent probes. Previously developed by Cravatt research group mapping of the ligandable proteome using fully functionalized enantiomeric probe pairs makes this field especially attractive for further investigations.
To support further research of the stereoselective interaction of proteins with chiral covalent small-molecules we designed and specially synthesized two sets of fragments with the most robust covalent warheads – acrylamides and chloroacetamides.
Typical Formats
Catalog No.
CEP-960-10-Y-100
Compounds
960
3 plates
Amount
10 µL of 100 mM DMSO solutions
Plates and formats
384-well echo qualified LDV microplates, 1,2 and 23, 24 columns empty, 320 compounds per plate
Price
Catalog No.
CEP-960-25-Y-20
Compounds
960
3 plates
Amount
25 µL of 20 mM DMSO solutions
Plates and formats
384-well microplates, Greiner bio-one, 1, 2 and 23, 24 columns empty, 320 compounds per plate
Price
Catalog No.
CEP-960-50-X-20
Compounds
960
12 plates
Amount
50 µL of 20 mM DMSO solutions
Plates and formats
96-well plates, 2D-barcoded microtubes with first and last columns empty, 80 compounds per plate
Price
Download SD files
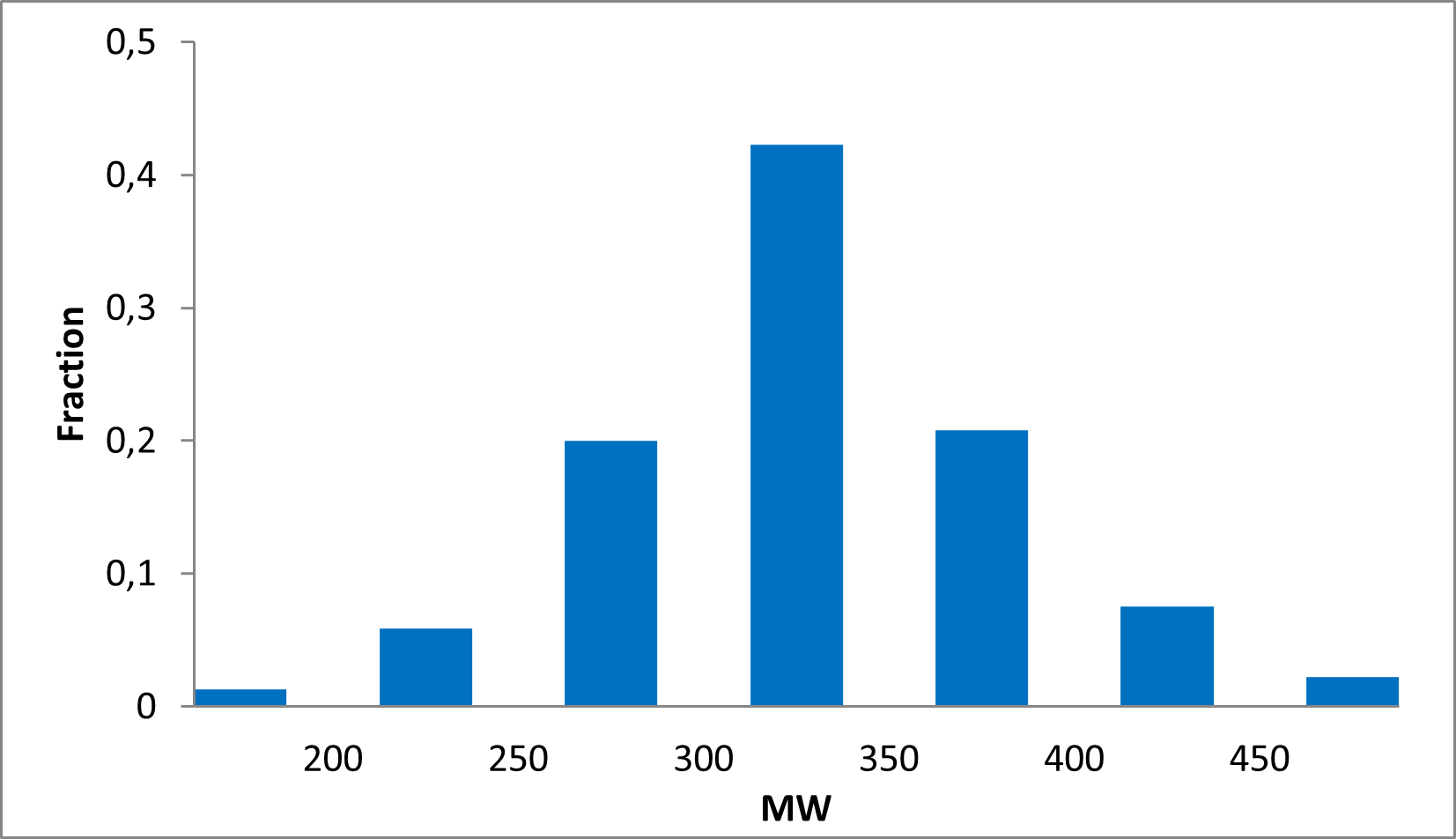
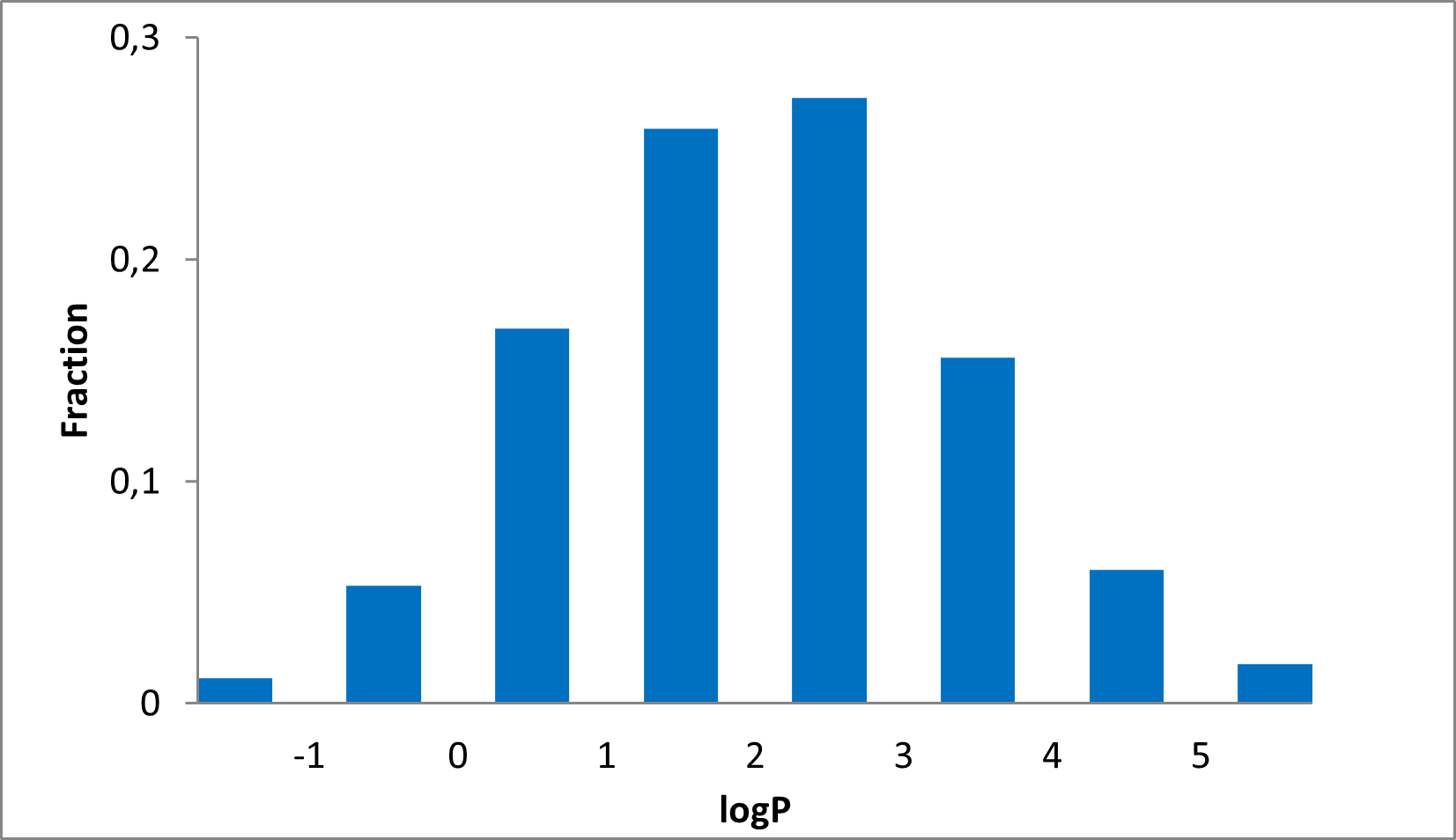
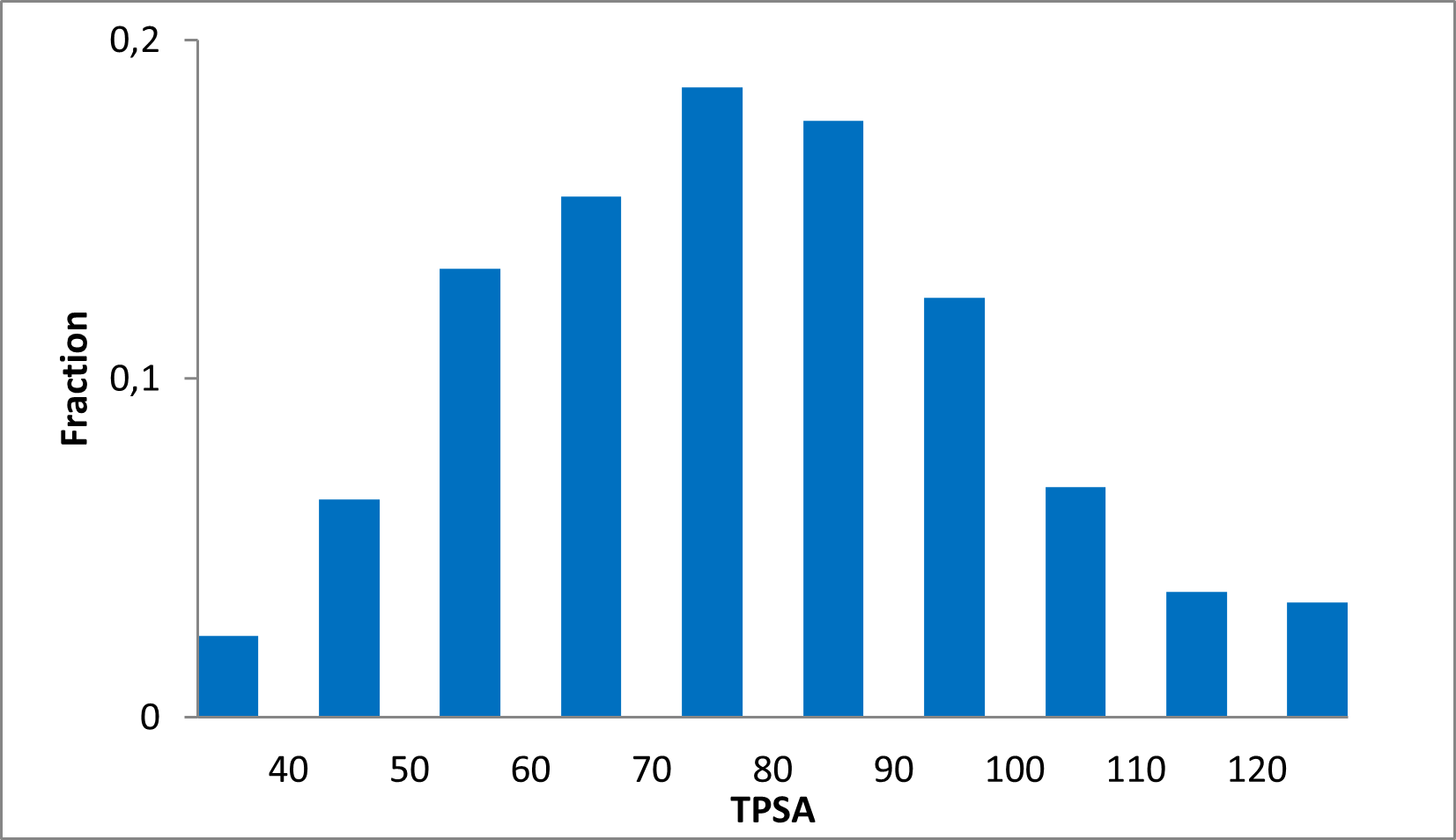
More than 6000 chiral building blocks with enantiomeric excess (ee) purity of 90%+ were analyzed and triaged for further synthesis of covalent binder pairs. The resulting set of over 2000 of the most diverse novel covalent modifiers has been synthesized. The enantiomeric purity of the corresponding enantiomeric pairs was analyzed by chiral chromatography. Compounds that passed rigorous QC and selection based on the diversity and Ro3 criteria were assembled into the Covalent Enantiomeric Pairs Library.
Key features
- Stereoselective interaction of target with enantiomeric covalent binder provides evidence of ligand-protein interaction
- Information about “correct” stereochemistry of hit on early stage
- Most common and well-validated warheads
Enatiomeric Pairs Fragment Library is plated at 100 mM concentration and is available for fast supply in different formats. The Library consists of two sublibraries, which can be acquired separately.
CNS-friendly molecules capable of BBB penetration
1 280 pre-plated fragments
Despite extensive research efforts, CNS drug discovery remains among the most challenging fields for drug developers. Factors such as heightened pollution, an aging population, and the impact of the global pandemic have collectively contributed to the increase of various CNS disorders. Regardless of great progress in diagnostics which is still limited in access, this field remains in desperate need of new effective, and safe treatments for most serious and frightening disorders.
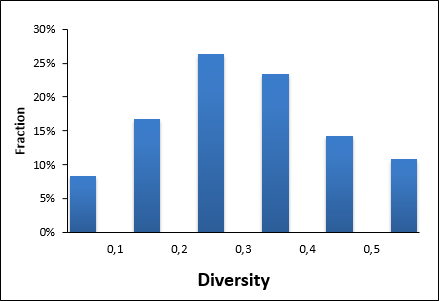
To help scientists at very early stages, we designed a specialized library of small fragments intended to bring convenient starting points that can quickly progress to potent CNS lead series. Analyzing all existing CNS drugs and drug candidates we created special selection criteria and structural patterns to identify potentially active molecules. This algorithm has been applied to Enamine’s Fragment Collection to reflect the most promising structures. The library was finalized with cluster-based diversity selections and manual review.
Typical Formats
Catalog No.
CNSF-1280-X-50-100
Compounds
1 280
16 plates
Amount
50 µL of 100 mM DMSO solutions
Plates and formats
96-well plates, first and last columns empty, 2D-barcoded vials (optional), 80 compounds per plate
Price
Catalog No.
CNSF-1280-Y-10-100
Compounds
1 280
4 plates
Amount
10 µL of 100 mM DMSO solutions
Plates and formats
384-well plates, 1, 2 & 23, 24 columns empty; 320 compounds per plate
Price
Catalog No.
CNSF-1280-Y-25-100
Compounds
1 280
4 plates
Amount
25 µL of 100 mM DMSO solutions
Plates and formats
384-well plates, 1, 2 & 23, 24 columns empty; 320 compounds per plate
Price
Key features
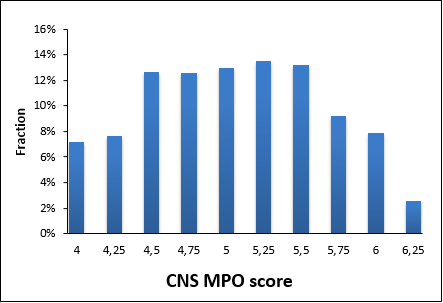
- High CNS scores: CNS MPO > 4.0 & QikProp CNS > 0
- Preferred scaffolds and chemotypes
- Rigorous MedChem filters and structure review
| Parameter | Range | Parameter | Range |
|---|---|---|---|
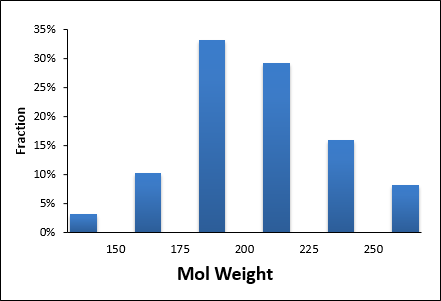
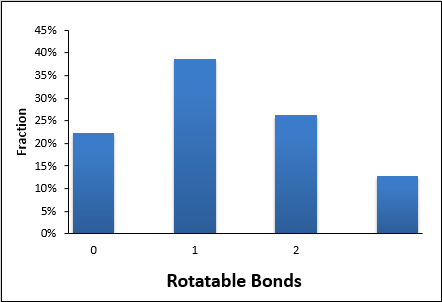
| MW | < 200 | RotBonds | ≤ 3 |
| ClogP | < 3.0 | ClogP-(N+O) | > -2.0 |
| PSA | ≤ 60 Å2 | Fsp3 | 0.15 ... 0.8 |
| Hb Donor | ≤ 3 | Ring count | ≤ 3 |
| Hb Acceptor | ≤ 5 | Basic Nitrogen | ≤ 2 |
| Total H-bonding | < 8 | S, Cl atoms | ≤ 2 |
| Carboxylic acids | ≤ 1, no more than 10% of CA | ||
| No more than 2 amide bonds, 87% with one amide bond | |||
| No quaternary Nitrogen, no NO2, Br, I, P | |||
Novelty
Compounds included into CNS focused Library were selected from all three screening collections - HTS, Advanced and Premium. Preference was given to the compounds synthesized within the last 4 years, based on innovative scaffolds and with use of advanced building blocks to enhance the novelty value of the library.
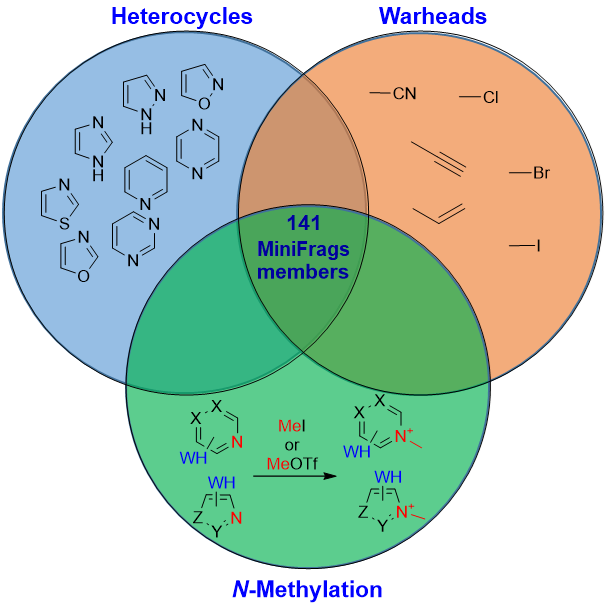
Covalent Heterocyclic Fragment Library for identification of Cryptic and Allosteric Pocket
141 compounds
This unique library of small heterocyclic electrophiles developed by the research group of Prof. György Keserű have been shown to be effective in finding tiny new binding pockets for different protein targets (MedChemComm 2019, Nat. Commun. 2020 and Pharmaceuticals 2022). The library combines the advantages of Astex’s MiniFrags in exploring unprecedented binding site with that of covalent binders in higher affinity and easier detection. It consists of six and five-membered heterocycles most abundant in drugs that are equipped with only one or two-atom covalent warheads. This makes it unique in contrast to those often used in covalent screening, much larger acrylamides and chloroacetamides which can significantly influence the binding mode of active molecules. Thus, applying the smallest possible covalent function helps to avoid promiscuity and keep the same recognition pattern of non-covalent scaffolds.
On the other hand, the electrophile-first approach proved to be an effective way of developing covalent drugs (Nat. Rev. Drug Discov. 2022). A number of covalent probes and drug candidates, including recent examples of Nirmatrelvir and Sotorasib have been developed using covalent screening techniques.
Covalent MiniFrags is a unique tool for searching of new binding pockets, elaboration of discovered hits and growing vector identification. The XChem facility at Diamond LightSource UK is a strategic partner in pioneering applications of Covalent MiniFrags.
Typical Formats
Catalog No.
CMF-141-5-Y-500
Compounds
141
1 plate
Amount
5 μL of 500 mM DMSO solutions
Plates and formats
384-well acoustic LDV plates, first two and last two columns empty
Price
Catalog No.
CMF‑141‑25‑X‑100
Compounds
141
2 plates
Amount
25 μL of 100 mM DMSO solutions
Plates and formats
96-well plates, Greiner Cat. No 650201, round (U) bottom, 1 & 12 columns empty, 80 compounds per plate
Price
Key features
- Most common nitrogen-containing heterocycles
- The smallest covalent warheads
- Experimentally characterized stability and intrinsic reactivity
- Evaluated on a number of targets
Covalent MiniFrags Library consists of 85 heterocyclic electrophiles and 56 N-methylated functional heterocycles. Both subsets are described in detail in scientific publications and are now available for your research.
Specially selected molecules to target bromodomains
15 360 compounds
Bromodomains are protein domains found in various proteins and are involved in the recognition of acetylated lysine residues on histone proteins. These domains are named after their ability to recognize the acetyl-lysine side chain, which has a similar shape to a bromide ion. Bromodomains are essential in regulating gene expression and chromatin structure, as acetylation of histones is associated with open chromatin and active gene transcription. Dysregulation of bromodomain-containing proteins has been implicated in several diseases, including cancer and inflammation. As a result, bromodomains have become a popular target for drug discovery efforts, with several small molecule inhibitors in development for cancer and other diseases.
We focused on the search of active compounds against the most important Bromodomain families: BET subfamily: includes BRD2, BRD3, BRD4, and BRDT bromodomains, which are characterized by an extended ZA loop that interacts with acetylated lysine residues on histones. GCN5-related subfamily: This subfamily includes the GCN5 and PCAF bromodomains found in histone acetyltransferases and have a shorter ZA loop than the BET subfamily. TAF1-like subfamily: TAF1 and TAF1L bromodomains are found in transcription factor TFIID and have an N-terminal extension that interacts with other complex subunits. ATAD2-like subfamily: ATAD2 and ATAD2B bromodomains are found in AAA+ ATPases and have a long ZA loop and a unique insertion between the second and third alpha-helices. Additionally, we run docking calculations for BPTF protein that also contains bromodomain.
Typical Formats
Bromodomain Library is available for supply in various pre-plated formats, including the following most popular ones:
Catalog No.
BRD-15-0-Z-10
Compounds
15 360
12 plates
Amount
≤ 300 nL of 10 mM of DMSO solutions
Plates and formats
1536-well Echo LDV microplates, first and last four columns empty, 1280 compounds per plate
Price
Catalog No.
BRD-15-10-Y-10
Compounds
15 360
48 plates
Amount
≤ 10 µL of 10 mM DMSO solutions
Plates and formats
384-well, Echo Qualified LDV microplates #001-12782 (LP-0200), first and last two columns empty, 320 compounds per plate
Price
Catalog No.
BRD-15-50-Y-10
Compounds
15 360
48 plates
Amount
50 μL of 10 mM DMSO solutions
Plates and formats
384-well, Greiner Bio-One plates #781280, 1,2 and 23,24 columns empty, 320 compounds per plate
Price
Catalog No.
Library & follow-up package
Plates and formats
BRD-15-10-Y-10 screening library 15 360 cmpds, hit resupply, analogs from 4.7M+ stock and synthesis from REAL Space
Price
*We will be happy to provide our library in any other most convenient for your project format. Please select among the following our standard microplates: Greiner Bio-One 781270, 784201, 781280, 651201 or Echo Qualified 001-12782 (LP-0200), 001-14555 (PP-0200), 001-6969 (LP-0400), C52621 or send your preferred labware. Compounds pooling can be provided upon request.
Download SD file
Library design
We used a structure-based approach, molecular docking calculations, as the main method for the library design. All available PDB structures for each of the bromodomain subfamilies were analyzed and extracted from PDB and PDBe. Unique "protein-ligand" complexes were selected for analysis. The following PDB structures were included in our study: BRD2 (4j1p, 4a9o, 5uew, 6ffe, 7l6d, 7l9j, and 7oe8), BRD3 (3s91 and 7l9l), BRD4 (7ajn, 7axr, and 7c2z), and BRDT (4flp, 7l9a, and 7mrg); PCAF (5fdz and 5fe5); TAF1 (5i1q, 6p38, and 7jjg); ATAD2 (6veo); and BPTF (5r4i, 6lu5, and 7lp0).
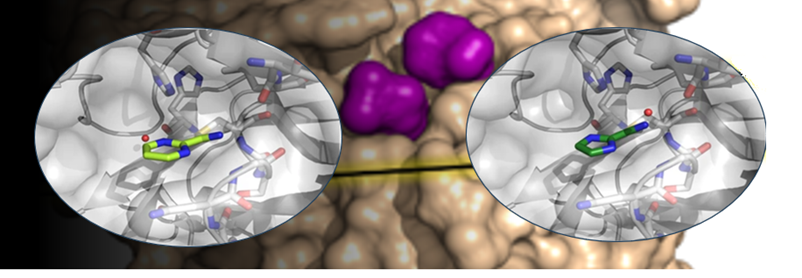
All simulations were performed with a common feature: the binding of a potential ligand to Asn inside the bromodomain binding pocket. All other binding points were dependent on the specific bromodomain and the structure of its native ligand. For example, in the left picture, the docking example of 7l6d is presented. The model is characterized by the following key features: (1) an h-bond acceptor to interact with Asn 429, an HOH molecule (which creates an h-bond bridge with Tyr 386), and an h-bond acceptor in the other part of the binding pocket to interact with the peptide backbone of Asp 377; (2) two aromatic groups to fill in the subpockets between Val 435, Leu 383, and between Pro 371, Leu 381; and (3) any atom group to fill in the subpocket among Tyr 428, Asn 429, and Leu 383. In contrast, in the right picture (5fe5), the model should contain slightly different binding properties: (1) an h-bond acceptor to interact with Asn 803, an HOH molecule (which creates an h-bond bridge with Tyr 760); (2) two aromatic groups to fill in the binding pocket and potentially create stacking interactions with Tyr 809.
Examples of molecular docking simulation to bromodomains
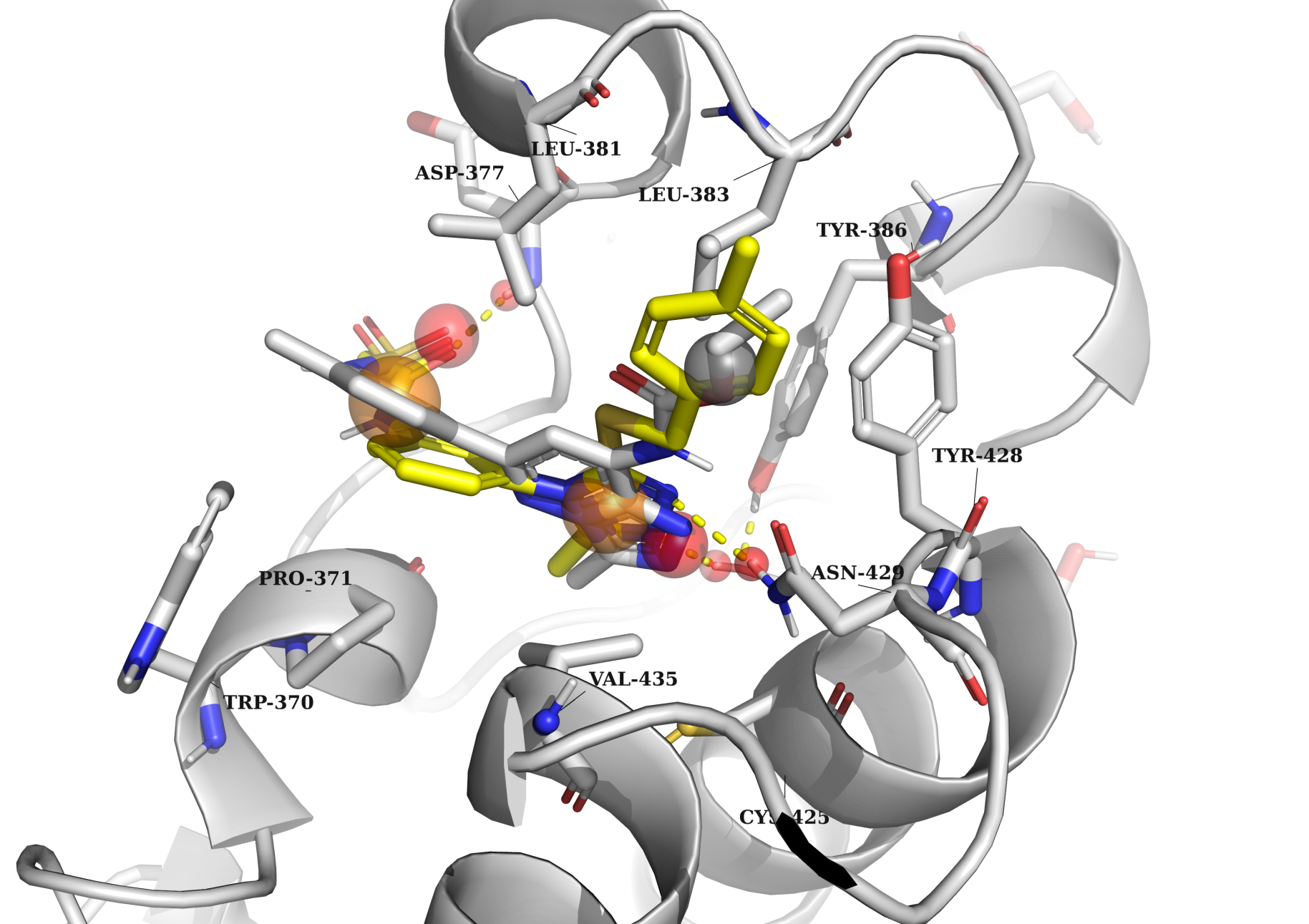
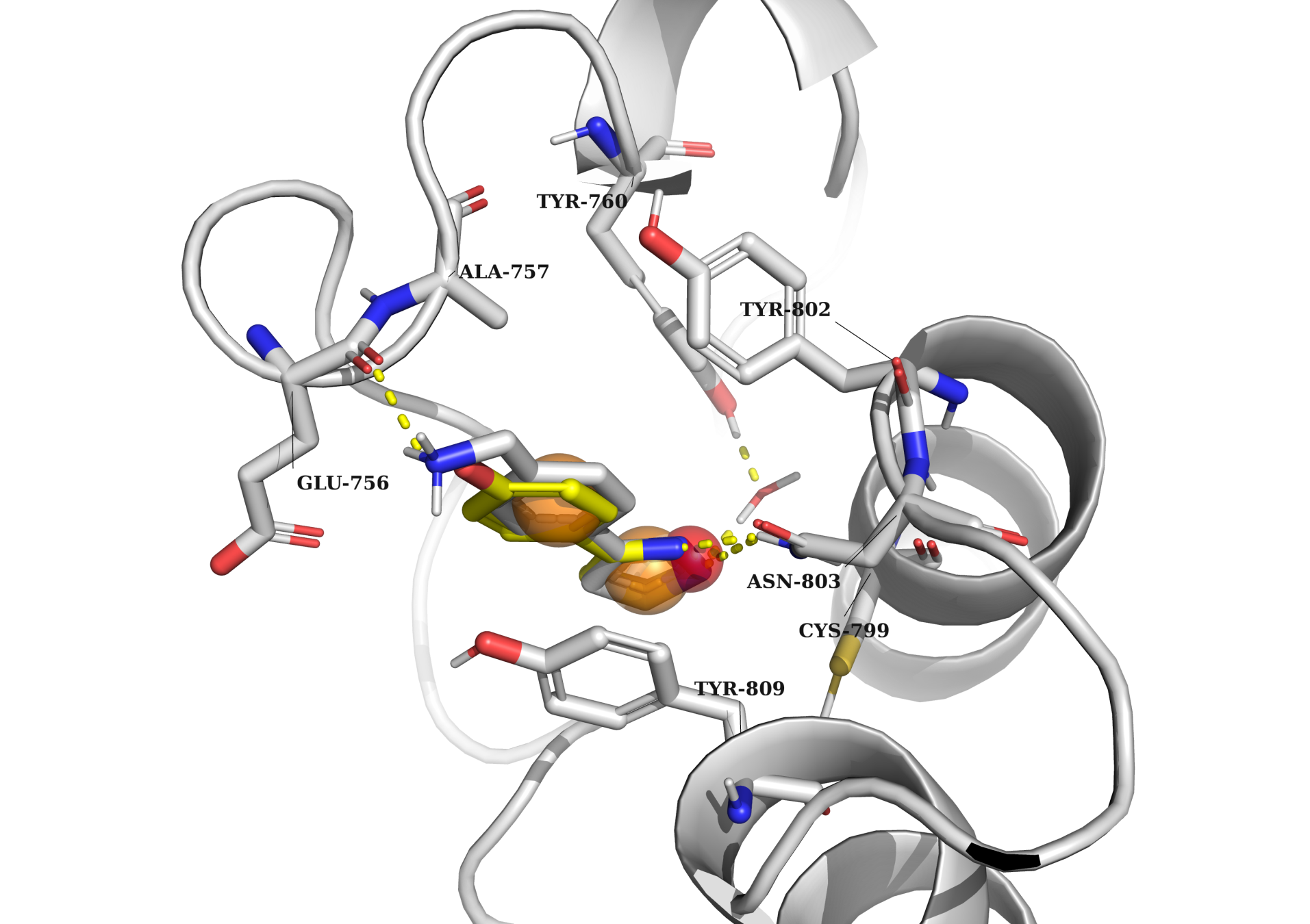
The molecular docking simulations to bromodomains were conducted using the following color scheme: red spheres represent h-bond acceptors, blue spheres represent h-bond donors, orange spheres represent aromatic groups, and grey spheres represent any other atom types. The protein and its native ligand are highlighted in grey, while an example of a docked ligand is shown in yellow.
Collection of referred small molecules with carefully collected activity data
2 405 compounds
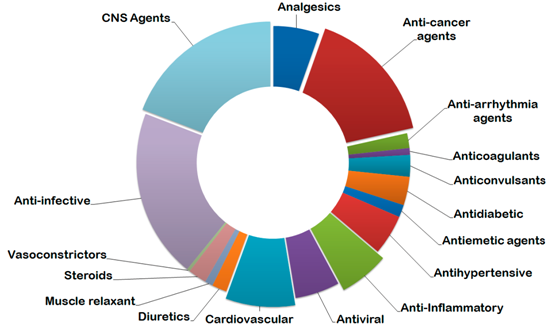
To address continuously growing interested to Drug Repurposing we designed and carefully collected a Bioactive Reference Collection of over 2 400 compounds with extensive target classes’ coverage and the broadest possible therapeutic areas applications – from CNS agents and anti-infectives to anticancer drugs and steroids.
Represented collection of carefully selected compounds includes 1 123 FDA approved drugs, as well as “tool compounds” with validated biological activity, active metabolites/prodrugs, and drug candidates that are currently undergoing clinical trials.
Download SD files
Bioreference Compound Library
Library code: BRL-2405
2 405 compounds
Version: 13 November 2023
FDA approved drugs
Library code: FAD-1123
1 123 compounds
sublibrary of Bioreference Library
Version: 9 November 2023
Bioactive Compounds
Library code: BAC-1282
1 282 compounds
sublibrary of Bioreference Library
Version: 14 November 2023
Ready-to-use, fully referred, alternative for compound screening.
Any compound from BRC collection can be ordered individually as focused sets: target-based or by therapeutic area.
Proven and reliable
- Stringent quality control using most advanced methods.
- Synthetic chemistry capacity with professionals in organic chemistry experienced in diverse synthetic methods and techniques.
- Drugs synthesis and their functional modifications for target identification and other purposes.
Distribution of compounds by therapeutic area
Related products & services
Target identification toolbox
Additionally we provide broad functionalization of Drugs and relative actives with covalent warheads, biotin or/and dye-linking, PED-derivatization and other modifications. Versatile chemistry and the largest stock of valuable reagents enables Enamine to produce new functionalization of well-known drugs.
We combined most diverse approaches and possible structural modifications to achieve most complete representation of our Target identification tools.