Designed for discovery of new Voltage-gated calcium channel blockers
10 560 compounds
Calcium ion channels are responsible for an unusually large variety of physiological functions. Calcium ions entering the cell through voltage-gated channels serve as the second messenger of electrical signaling, initiating many different cellular events. As a target class calcium channels offer both challenges first of in designing selective antagonists of the channel subtypes and great opportunities following proof-of-concept provided by the marketed drugs.
Enamine voltage-gated targeted library encompasses both known chemotypes and molecules based on novel scaffolds identified in our in silico studies and MedChem based scaffold hopping projects.
Download SD file
Main futures of the library:
- Lead-likeness: 86%
- Average Fsp3 0.38; average TPSA 60.5 Å2
- Novel Chemotypes
Library design
We have undertaken in-depth analysis of known calcium channel blockers and carefully selected 10 560 most promising drug-like structures from our screening collection exceeding 4.7M+ small molecular weight compounds.
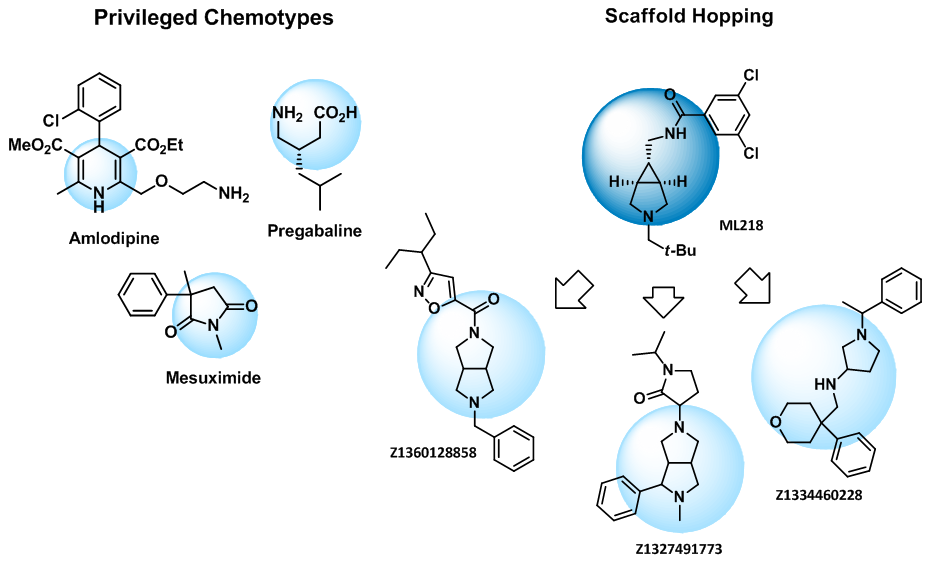
Privileged chemotypes such as dihydropyridines, GABA-analogues, succinimides, 4-substituted prolines, and quinazolines were included in the library.
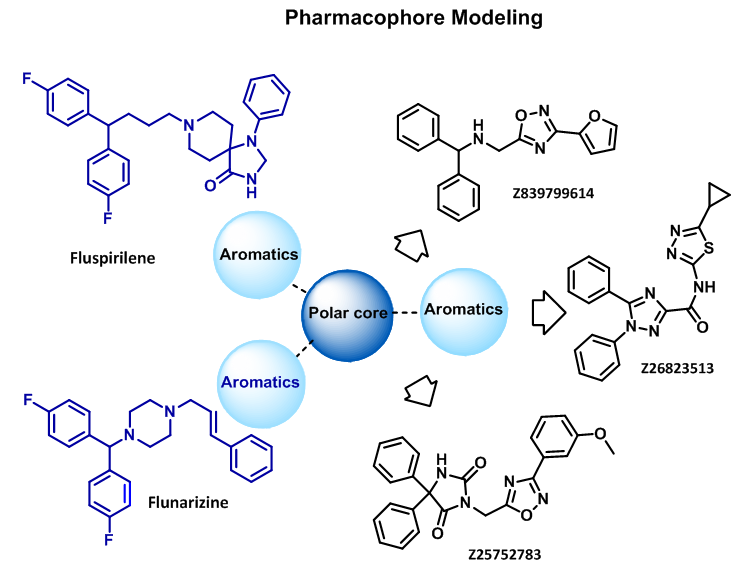
Pharmacophore modeling was carried out for several relevant calcium channel blockers resulting in about 1 700 compound set.
Scaffold hopping Structures of numerous known calcium channel blockers contain piperazine or 4-aminopiperidine. Recently several successful examples of bioisosteric replacement of these motifs by other diamine cores have been reported. We have extended this approach to a rich variety of compounds built on unique analogues of piperazine and 4-aminopiperidine available exclusively from Enamine and continued with other cores, e.g. imidazopiredazines, providing novel sets of compounds.
Molecular properties Drug like compounds with the most attractive structural and physical chemical properties. HBond Donors 0… 4, HBond Acceptors: 1 … 8, Rotatable Bonds: 0 ... 10, TPSA 3 … 140 Å2



