Most diverse linkers for your discovery program
Bifunctional Molecules such as PROTACs, DUBTACs, LYTAC, AUTOTAC, GlueTAC, CHAMP, and others are among the hottest new modalities in modern Drug Discovery. These molecules can be considered as two actives connected with linkers. Numerous scientific studies suggest that the linker’s length, polarity, and rigidity can play a crucial role in a tertiary complex formation, its stability, and the pharmacokinetic (PK) properties of the bifunctional molecules. Quick and easy access to various linkers is essential for a rapid “Linkerology” search since it is next to impossible to predict a “perfect linker” for a given target. Vast majority of protein degraders were developed through the most empirical optimization of linker composition, often driven by the commercial availability of PEG- and carba-precursors.2 As Derek Lowe mentioned in his post on Linkerology: “The only thing we can say for sure now, is that it's a complex situation with a lot of room for surprises. Go make some more linkers!”
Being the largest supplier of Building Blocks, Enamine provides a wide assortment of linker molecules from stock. They can be used for the rapid design and synthesis of different bifunctional molecules. Fine adjustments can also be made by searching over 5 million MADE Building Block linkers.
Download SD files
Linear linkers
These are classical tools that are often applied for the design and investigation of new PROTACs, DUBTAGs, and other bifunctional molecules. Enamine offers a diverse collection of building blocks for the construction of linear PEG- and carba-linkers with different lengths, tail functional groups (FGs), and composition. This collection is continuously enriched with new compounds from our synthesis program.
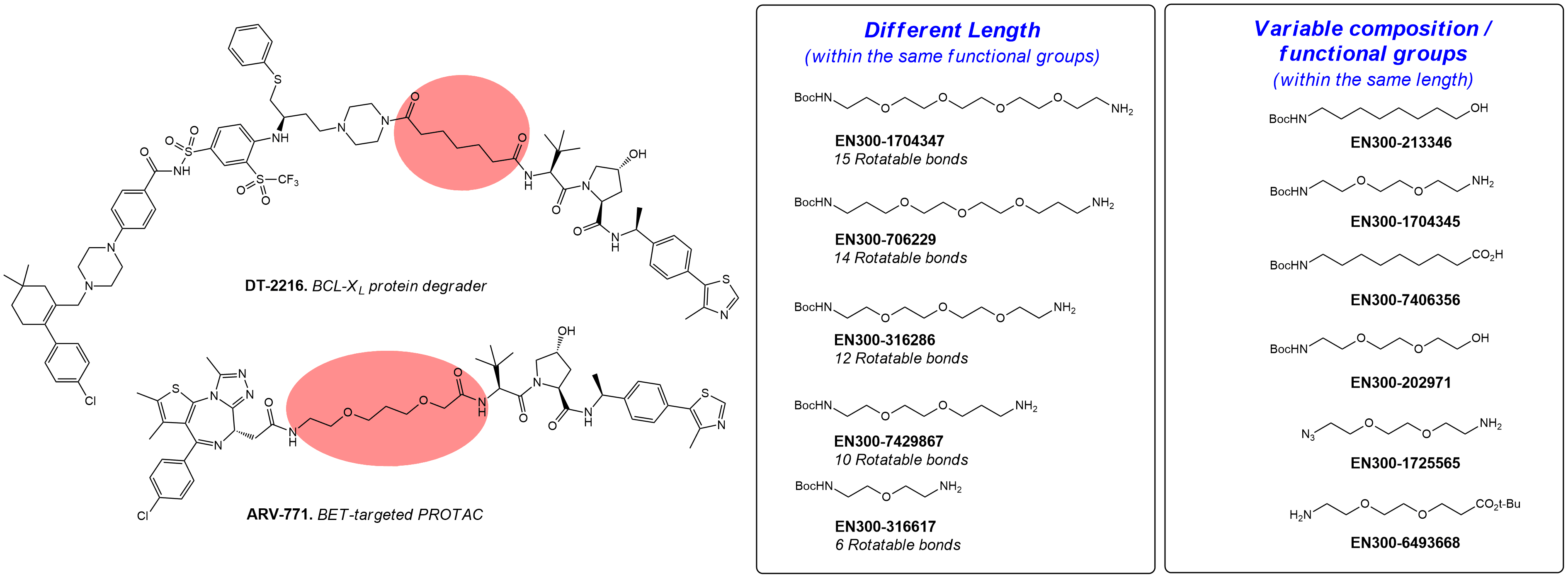
Examples of PROTACs with linear linkers.
Advanced linkers
More complex, with strained structure linkers have become popular in the last couple of years. These advanced linkers contain in their structure cycles, spirocycles, double and triple bonds, and multiple heteroatoms.
Based on the literature data, we singled out the following general criteria for this linkers type: 1-3 cycles, maximum one aromatic/heteroaromatic ring, presence of end functional groups (with or without protection), absence of side functional/reactive group/motifs. The corresponding linker Building Blocks are available from our stock. We can also readily synthesize any other linker molecules upon your request.
Mono-Boc diamines
Mono-Boc diamines are the most popular linkers in the design of bifunctional molecules (first of all PROTACs). Various diamine linkers can be found in the structures of the drug candidates such as ARV-110, ARV-471, CFT8634, and FHD‑609. Recently Janssen chemists have demonstrated a straightforward Direct-to-Biology (D2B) approach to new protein degrader discovery starting from diverse N-Boc diamines.1 Enamine provides an outstanding collection of diverse diamines (by flexibility/distance/shape/polarity) which are useful for linkerology studies of new bifunctional molecules.
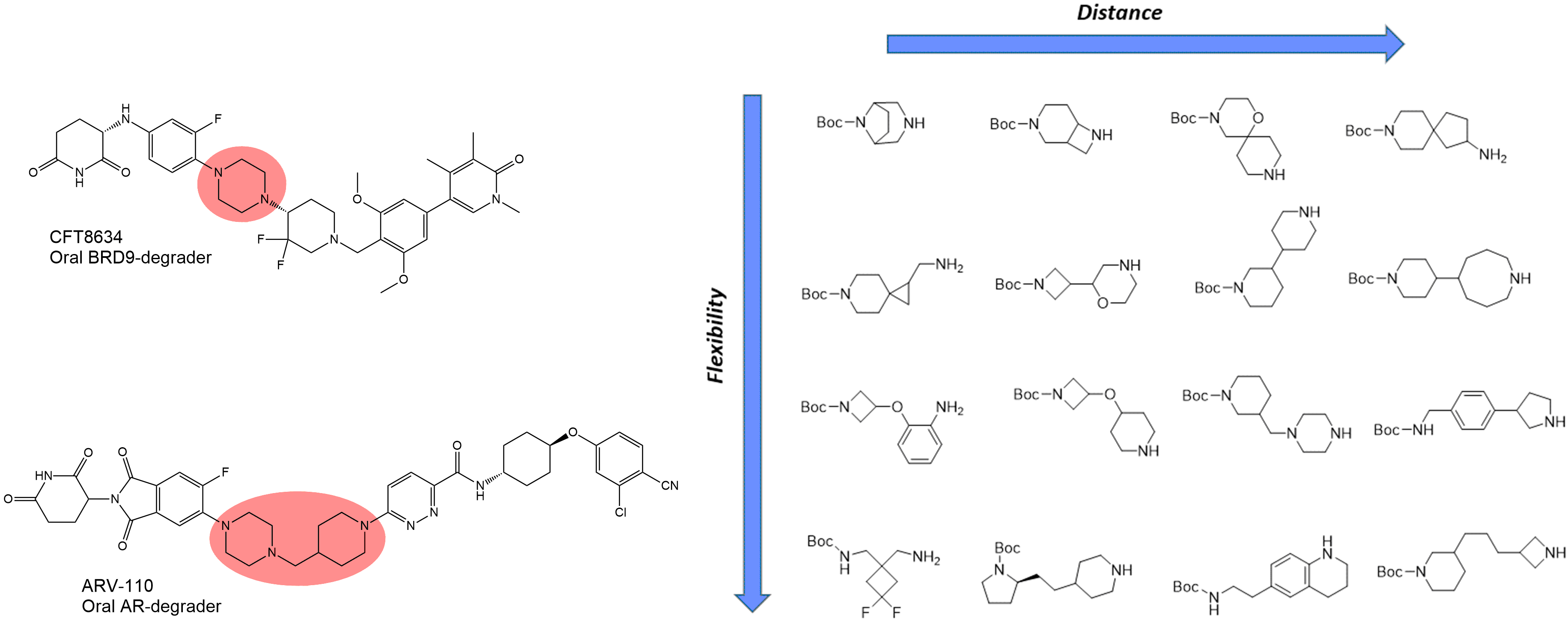
Other linkers
Besides diamines other linkers with diverse end-functional groups are widely used in the design of bifunctional molecules. Enamine provides various linkers with different combinations of functional groups (e.g., OH/halogen-acetylenes, amino acids, aminoacetylenes, aminoazides). Upon request, we can propose a set of linker Building Blocks according to your criteria.
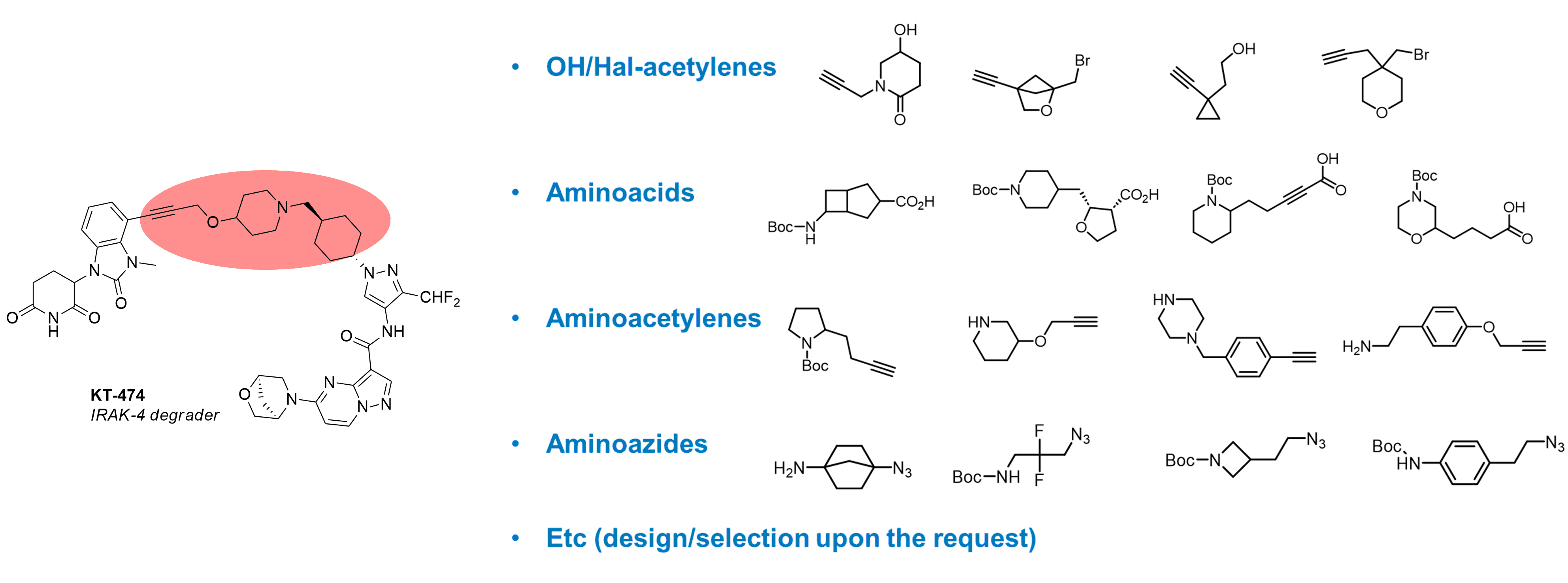
Selected publications
-
Direct-to-Biology Accelerates PROTAC Synthesis and the Evaluation of Linker Effects on Permeability and Degradation.
ACS Med. Chem. Lett. 2022, 13, 7, 1182–1190. DOI: 10.1021/acsmedchemlett.2c00124 -
Current strategies for the design of PROTAC linkers: a critical review.
Explor. Target Antitumor Ther. 2020, 273-312. DOI: 10.37349/etat.2020.00018



