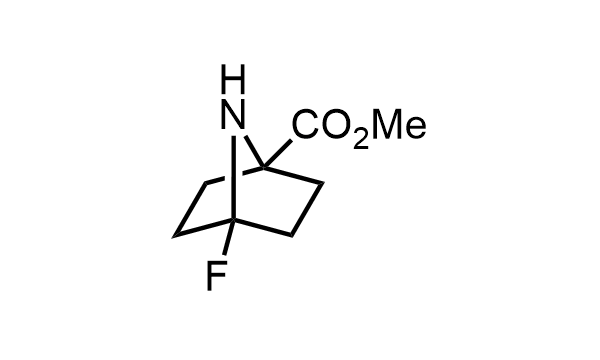
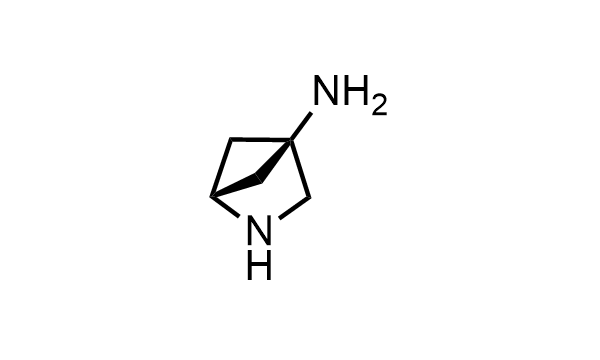
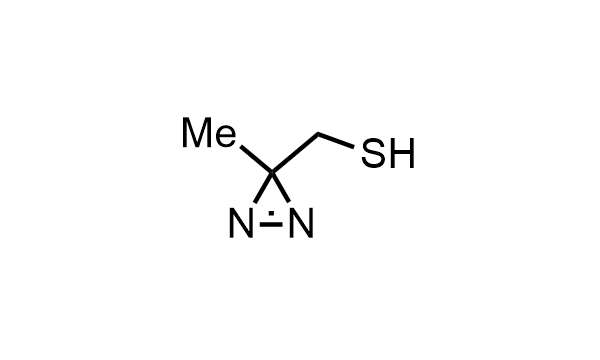
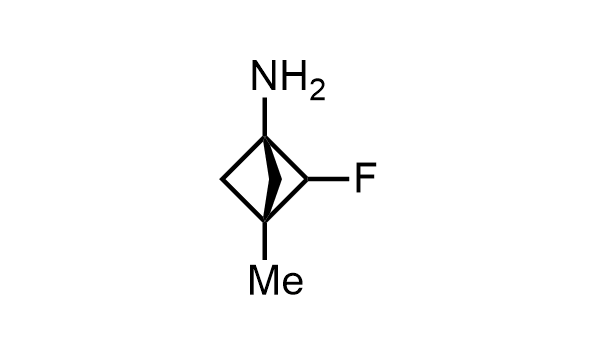
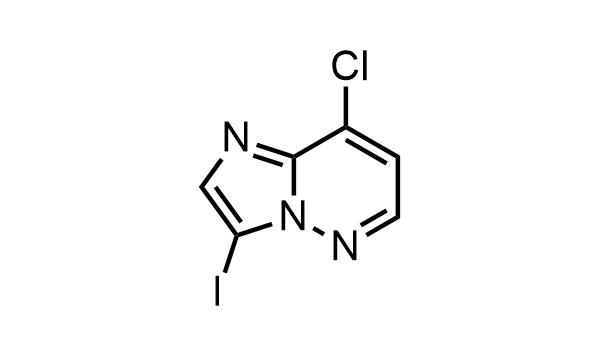
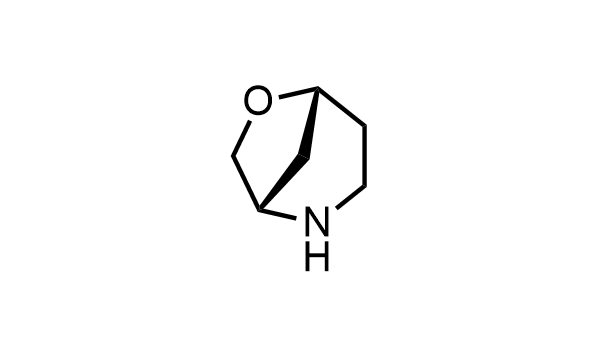
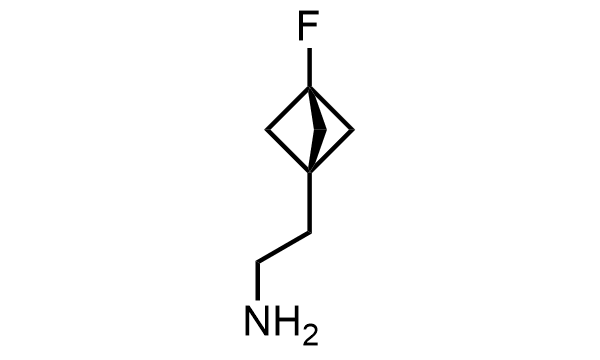
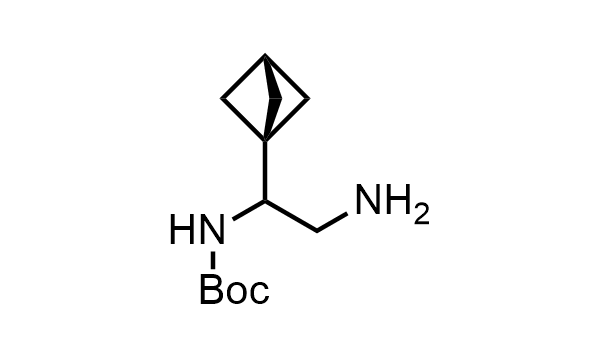
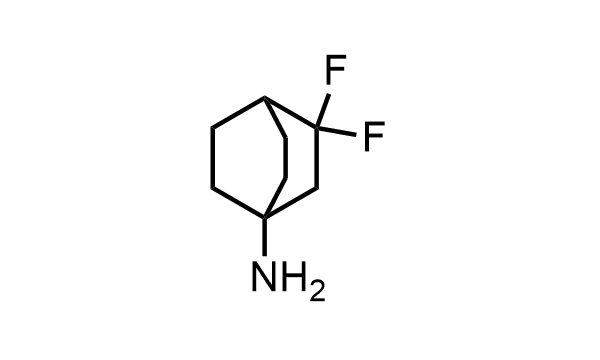
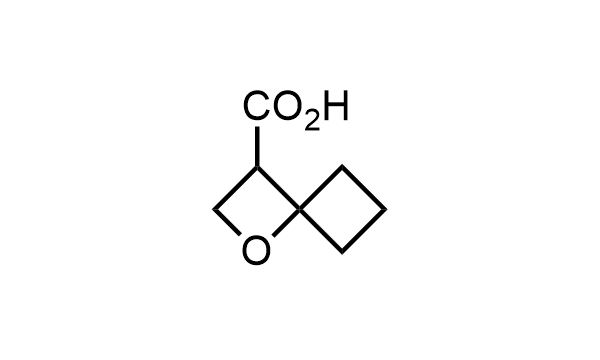
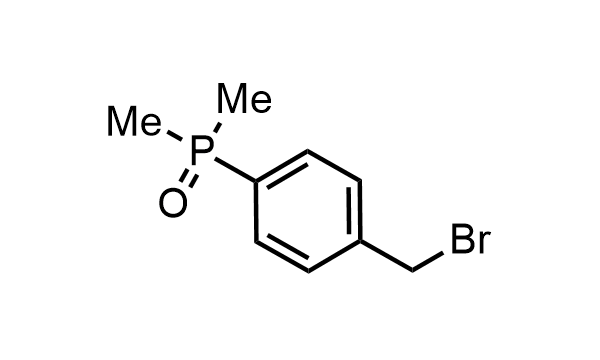
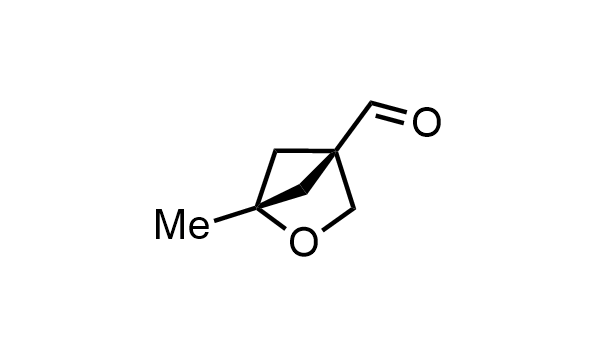
2 000 new building blocks are synthesized monthly. Here is an important update to our MedChem Highlights from May 2026
Recent News
-
13 May 2026 Press Release
Leveraging Public Infrastructure for Private Innovation: Enamine and PAL Align
Collaboration leverages PAL’s public research facilities and Enamine’s compound libraries and fragment progression support to accelerate fragment-based lead discovery (FBLD).
-
20 April 2026 Press Release
Enamine Strengthens Its Sustainable Procurement Program in 2026
In 2026, Enamine’s Sustainable Procurement Program is reaching a new level of maturity and impact. Building on many years of close cooperation with suppliers – and reinforced by global best practices – the Company is further strengthening its approach to responsible and sustainable procurement across the entire supply chain.
-
07 April 2026 Press Release
Chemspace, Enamine, and Paul Scherrer Institute Launch a Joint Service for Fragment-Based Lead Discovery
Chemspace, a drug discovery engine providing machine learning-guided support for hit identification, hit to lead, and lead optimization using the largest purchasable collection of chemicals and biologics, Enamine, a leading provider of small molecules and drug discovery services, and the Paul Scherrer Institute (PSI), the largest research institute for natural and engineering sciences in Switzerland, have announced today a strategic collaboration aimed at creating an efficient platform for fragment-based drug discovery in the ultra-large chemical space.
Upcoming events
Request product information
Please leave your request for any product you wish. We will be happy to search it for you in our databases or offer its custom synthesis