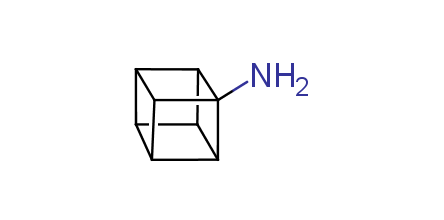
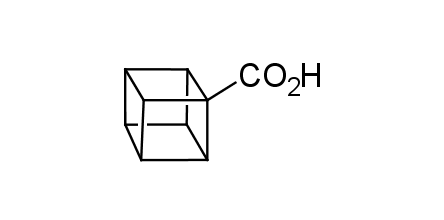
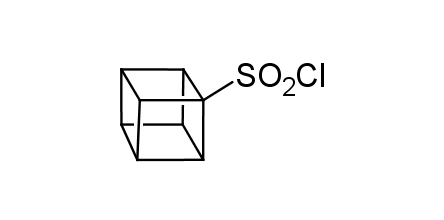
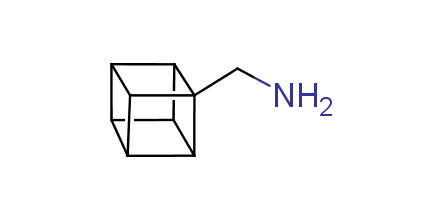
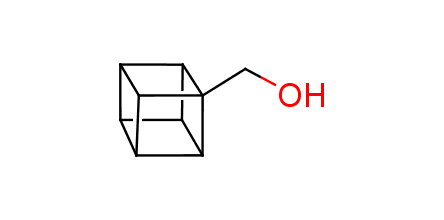
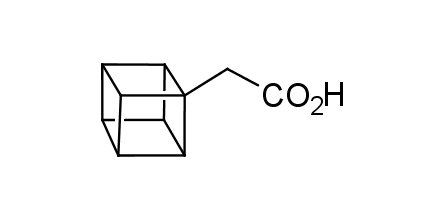
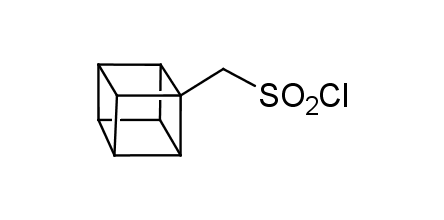
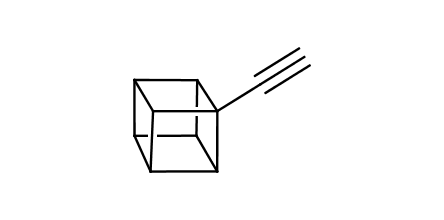
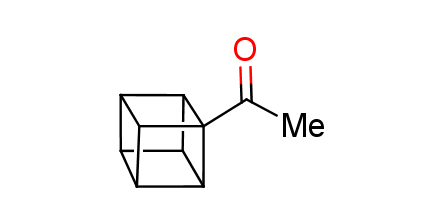
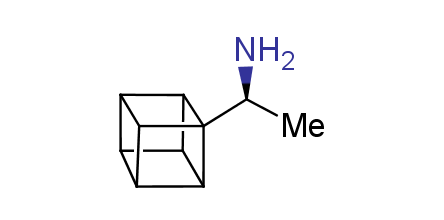
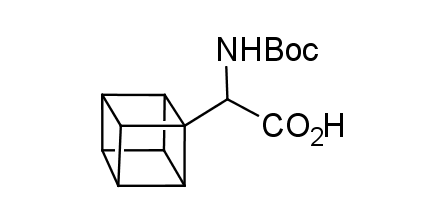
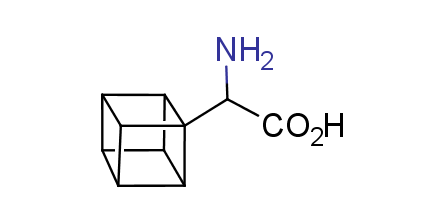
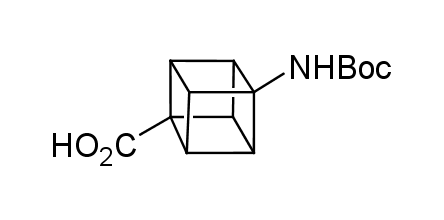
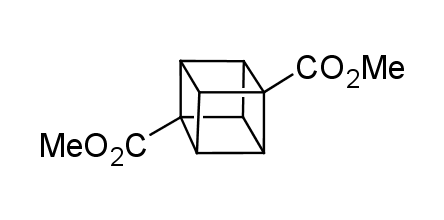
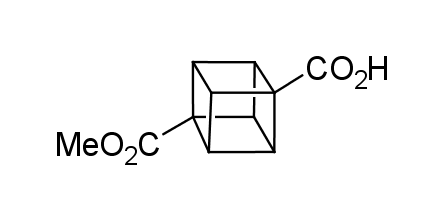
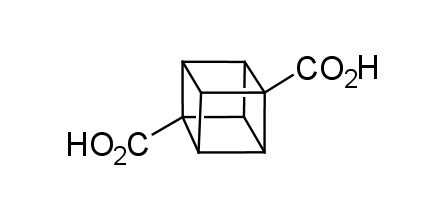
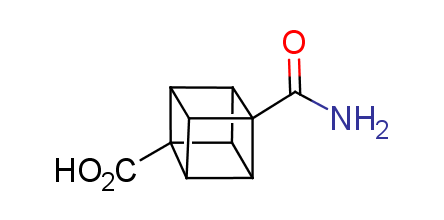
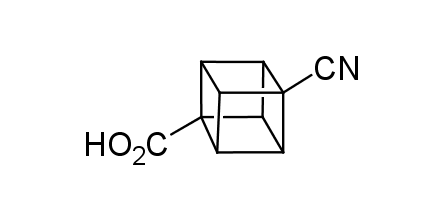
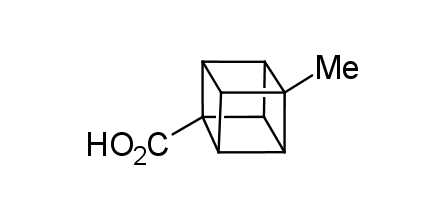
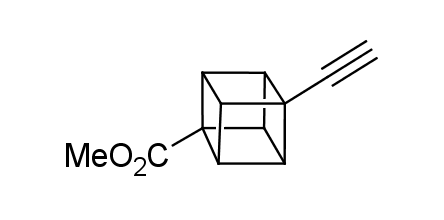
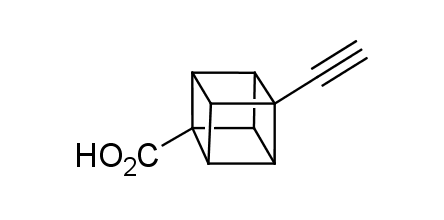
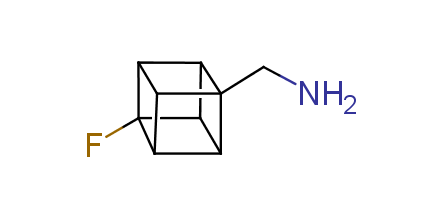
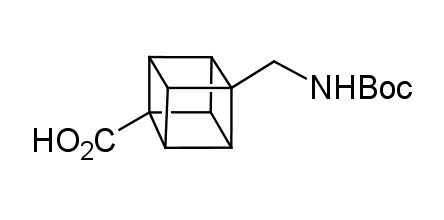
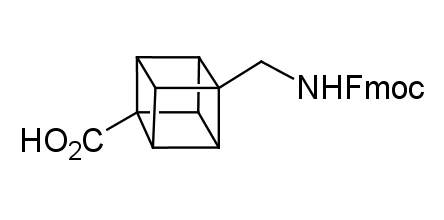
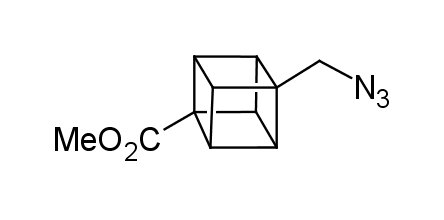
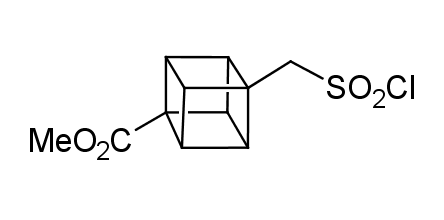
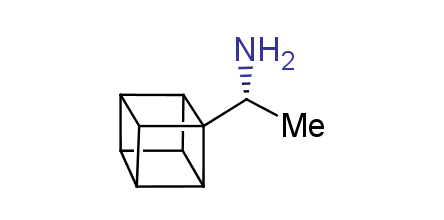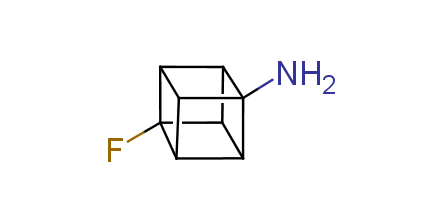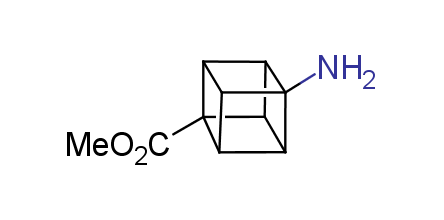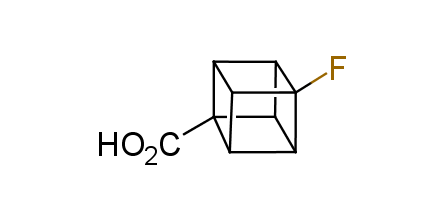In 1992, Eaton predicted a high potential of cubane in a pharmaceutical research as a bioisoster of benzene, based on their similarity in size. In 2016, Williams and collaborators showed that replacing a benzene ring in the neurotropic compound Leteprinim with cubane beneficially affected activity and solubility of the parent compound. The cubane analogue significantly outperformed pesticide Diflubenzuron. Since then the cubane-containing building blocks have been playing an important role in medchem projects, as mimics for the para-substituted benzene ring. In this context, Enamine offers a library of cubane-containing building blocks for drug design.
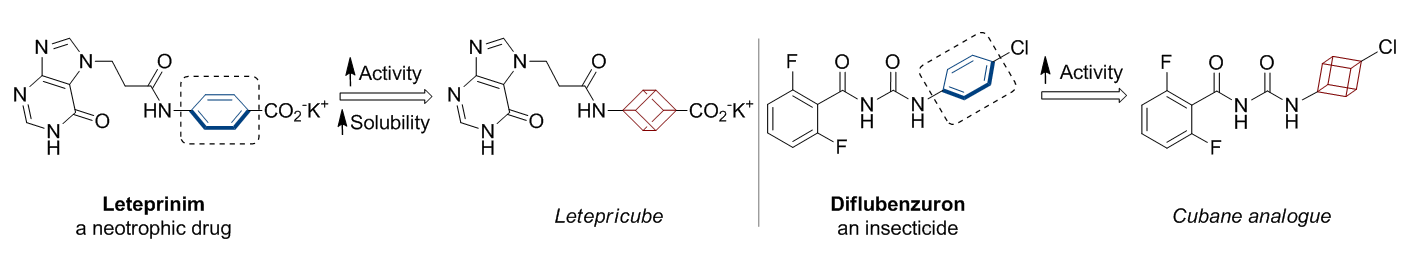
Concept

We offer:
Cubane-containing building blocks from stock on a 5-10 g scale.