6 119 compounds in stock and over 1.1 million in REAL
960 compounds pre-plated Sulfonyl Fluoride Fragment Library
640 compounds pre-plated Sulfonyl Fluoride Screening Library
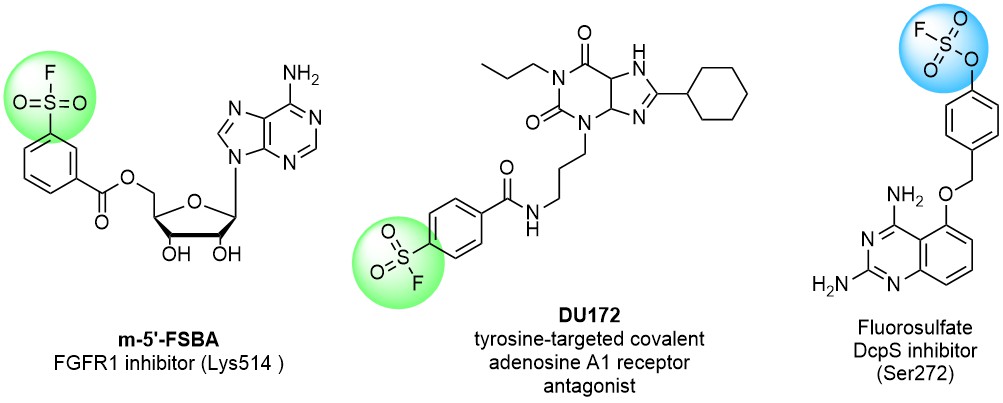
Sulfonyl fluorides are becoming increasingly popular in covalent probe discovery and are often used as tool compounds in chemical biology. Their high stability and low reactivity made them promising in modern drug discovery. Sulfonyl fluorides are known to react with multiple nucleophilic amino acid residues including Tyr, Lys, His, Arg, Ser and Thr. That is especially important due to the much higher abundance of Ser and Lys in proteins over Cys (on average about 9% and 6% vs 2%).
Download SD files
6 119 compounds for cherry-picking
Plated libraries in stock:
There is a significant difference in reactivity between fluoro sulfonates, sulfamoyl fluorides, and alkyl aryl/hetaryl sulfonyl fluorides, which enables the design of selective covalent binders. For example, less reactive fluoro sulfonates have been reported to react better with Tyr phenolic hydroxy groups than sulfonyl fluorides and sulfamoyl fluorides.
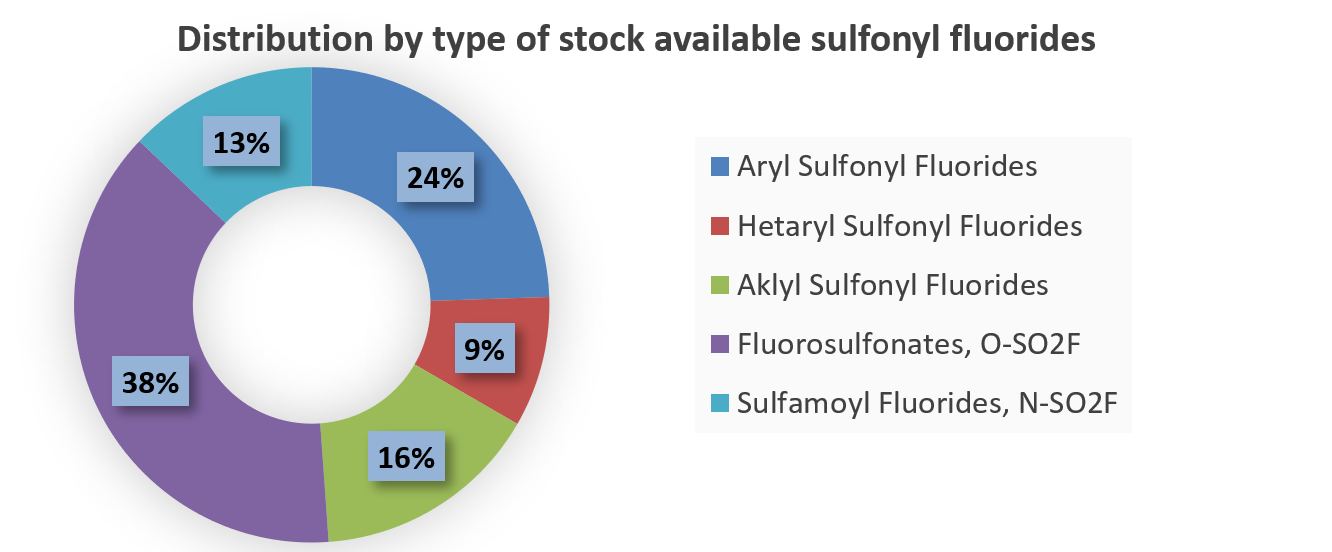
A recently published study by the research group at GSK on hydrolytic stability and reactivity of various sulfonyl fluorides, reveals interesting findings and a way to profile sulfonyl fluorides. Several substituted benzene sulfonyl fluoride scaffolds have been reported to possess balanced reactivity due to enough stability to hydrolysis and moderate activity toward nucleophiles.
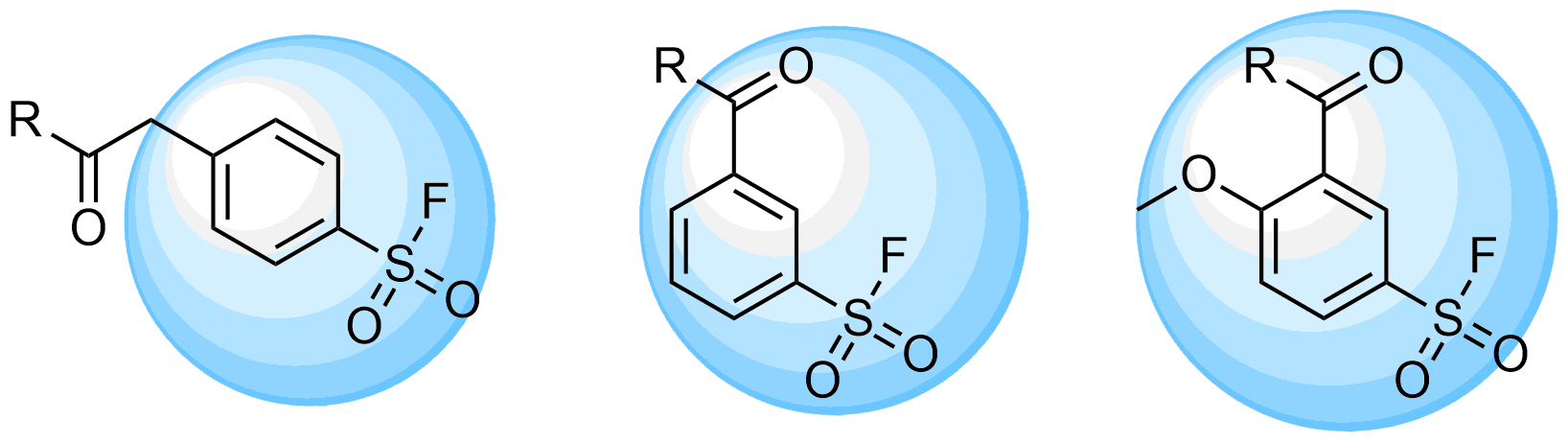
REAL Sulfonyl Fluorides
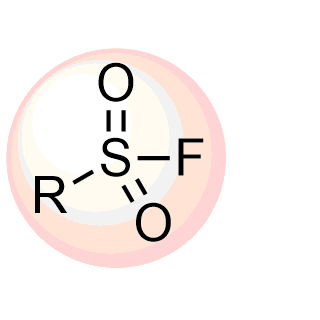
Over 701k REAL Sulfonyl fluorides, including aryl, hetaryl and alkyl sulfonyl fluorides
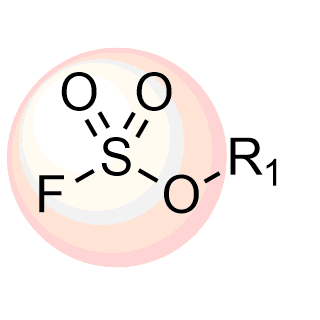
Attractive REAL Fluorosulfonates, over 94k compounds available for fast parallel synthesis with at least 80% success rate
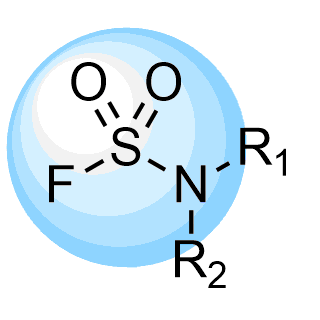
REAL Sulfamoyl fluorides, 210k for fast parallel synthesis within 3 weeks only
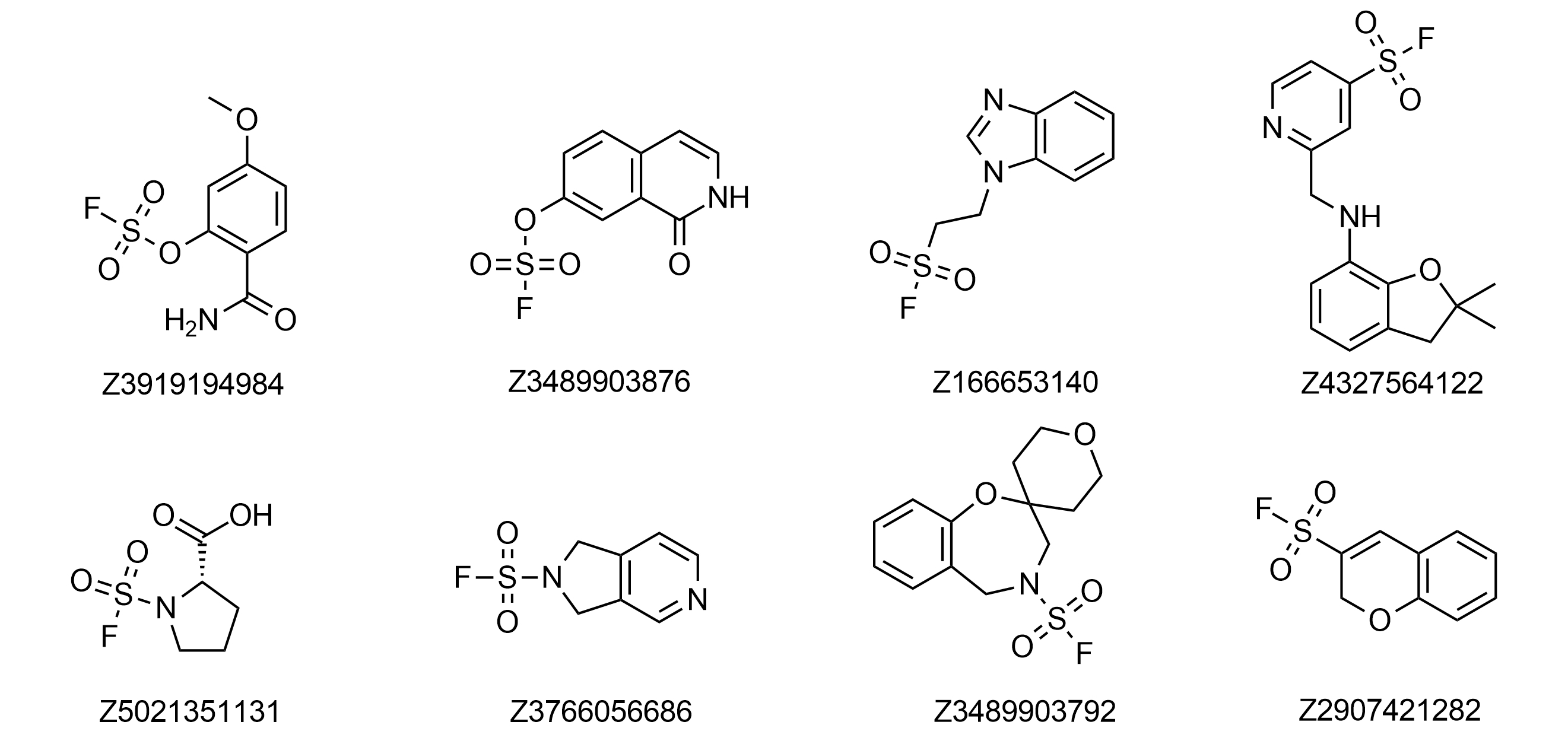
Examples of pre-plated Sulfonyl Fluorides
Covalent Screening Library: 640 Sulfonyl Fluorides plated at 10 mM in DMSO
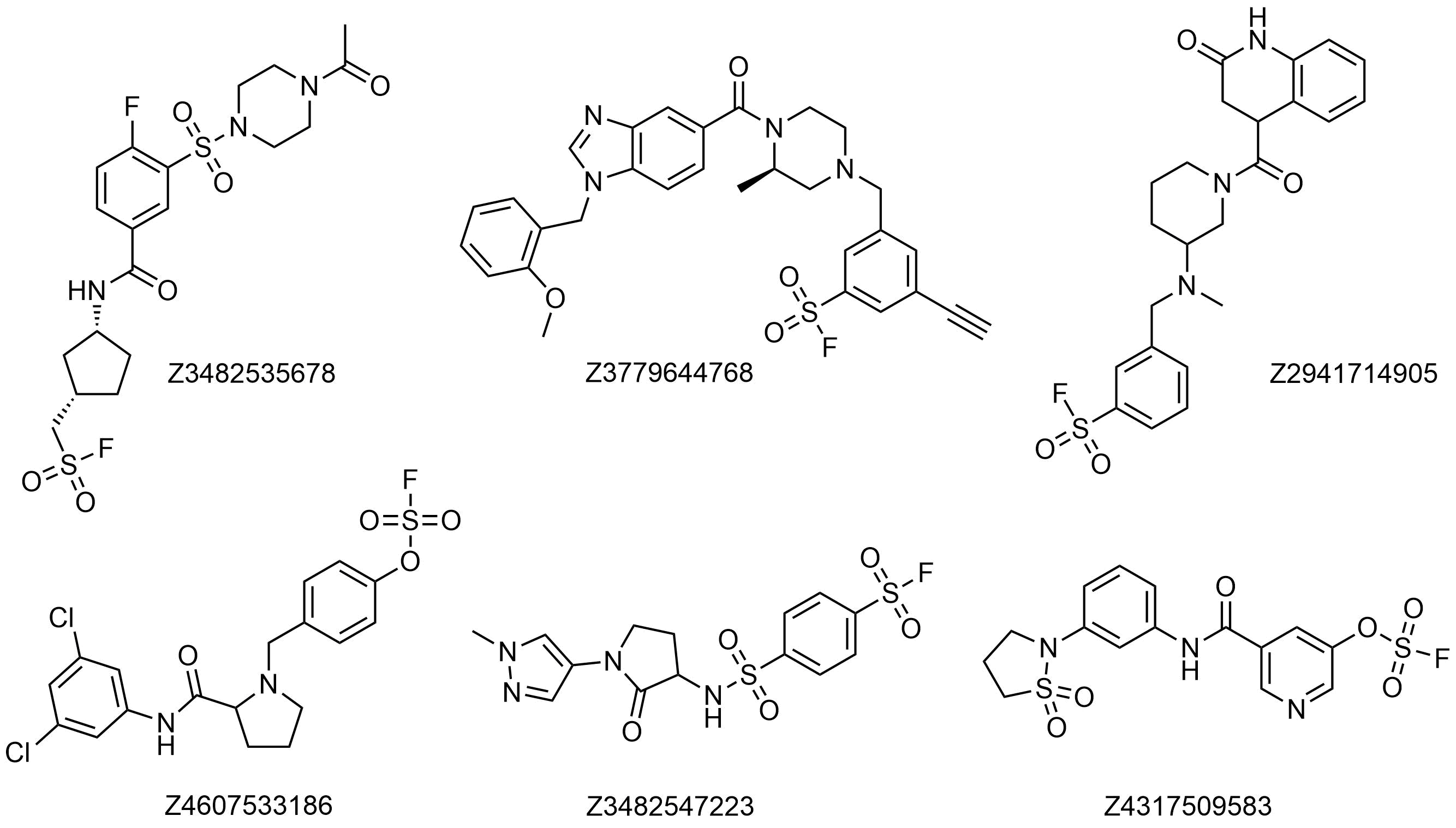
Covalent Fragment Library: 960 Sulfonyl Fluorides plated at 100 mM
For easy and efficient exploration of novel protein targets and proteomics screening platforms
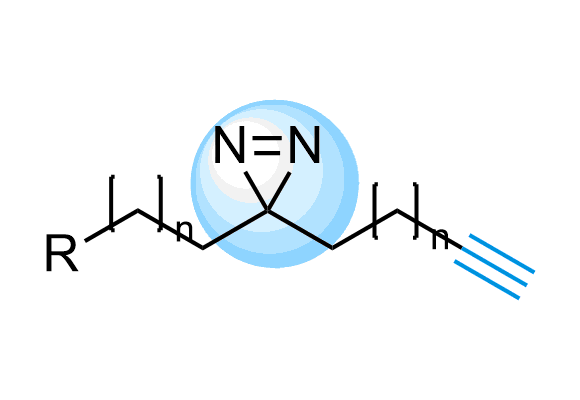
Photoaffinity labeling (PAL) is a successful and widespread strategy for ligand-binding site mapping, protein target-fishing, and target identification with affinity-based probes (AfBP). Medicinal chemists and structural biologists often use this powerful technique to reveal molecular targets after phenotypic screens and investigate signaling pathways and interactions between proteins of interest. PAL has become one of the most used tools in chemical proteomics and the studies of protein-protein interactions (PPIs).
Diazirines, benzophenones, and aryl azides are traditional, well-described photocrosslinkers (PLs) used in PAL. Upon photoirradiation, these moieties produce highly reactive species that quickly react with adjacent protein parts resulting in covalent binding, which can be further identified by mass-spec analysis and other methods. We at Enamine have been working on synthesizing new PLs containing molecules for years and now offer the largest and most diverse source of Photoaffinity Compounds, including a vast collection of Fully Functionalized Compounds.
Download SD files
Fully Functionalized Compounds
The use of diazirine in probe molecules, together with the functional acetylene group, allows screening directly in cells. Originally described in the Cell paper2 by Ben Cravatt, this approach has been adopted by other research groups for several successful projects, including ‘direct-to-biology’ high-throughput chemistry screening platform reported by GSK researchers.
We offer over 6 000 Fully Functionalized Compounds from stock and more than 100 million REAL compounds.
Download Fully Functionalized compounds in stock
Download REAL Fully Functionalized Compounds
Browse our building blocks
Trifluoromethyl Diazirines
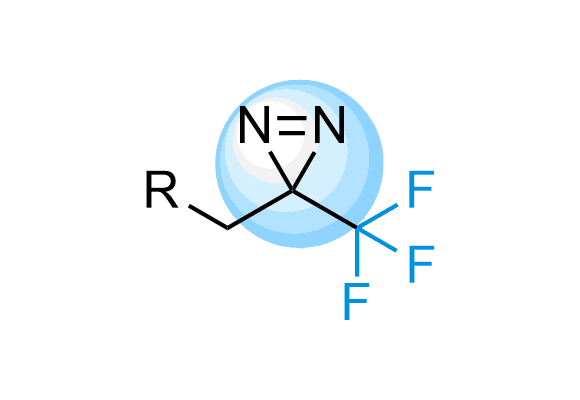
Trifluoromethyl diazirines work more selectively on target because the isomeric diazo compounds formed as side products upon photoirradiation are relatively unreactive and do not largely interfere. We offer over 700 Trifluoromethyl diazirine compounds from stock to select from and more than 300 thousand REAL compounds that we can synthesize on demand using our off-the-shelf CF3-diazirine Building Blocks.
Download ready Trifluoromethyl Diazirine compounds
REAL CF3-diazirine photoaffinity ligands
Benzophenones
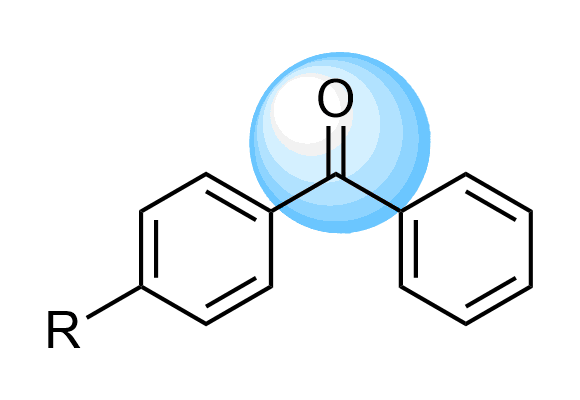
We offer over 5 000 Benzophenone Compounds from stock and will be happy to help you with the design and synthesis of new derivatives.
Aryl Azides
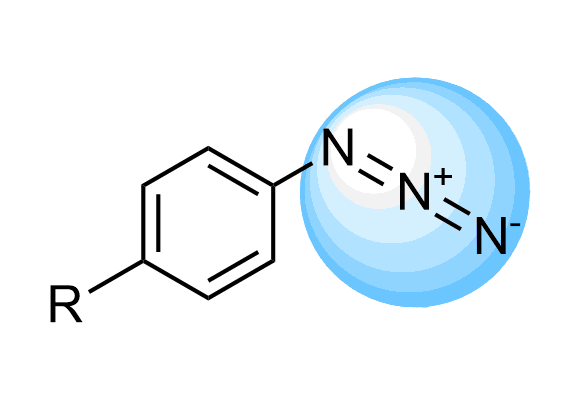
We offer over 50 Arylazides from stock with an extensive array of novel azides that can be synthesized on demand based on our massive collection of azide building blocks.
Synthesis
We have refined our parallel chemistry protocols to efficiently synthesize Photoaffinity Compounds using our own collection of photoaffinity building blocks. This collection is the largest in the world, providing access to all classes of crosslinkers having a wide diversity of functional groups to use in synthesis. Over 15 000 ready-to-use Photoaffinity Compounds have been already synthesized. You can additionally select from over 1 million REAL Compounds bearing photoaffinity warheads. These compounds will be synthesized within just 3-4 weeks with a high (80%) success rate. We also offer synthesis of any bespoke compounds bearing specific photoaffinity moieties attached to selected backbones as well as compound libraries virtually of any size.
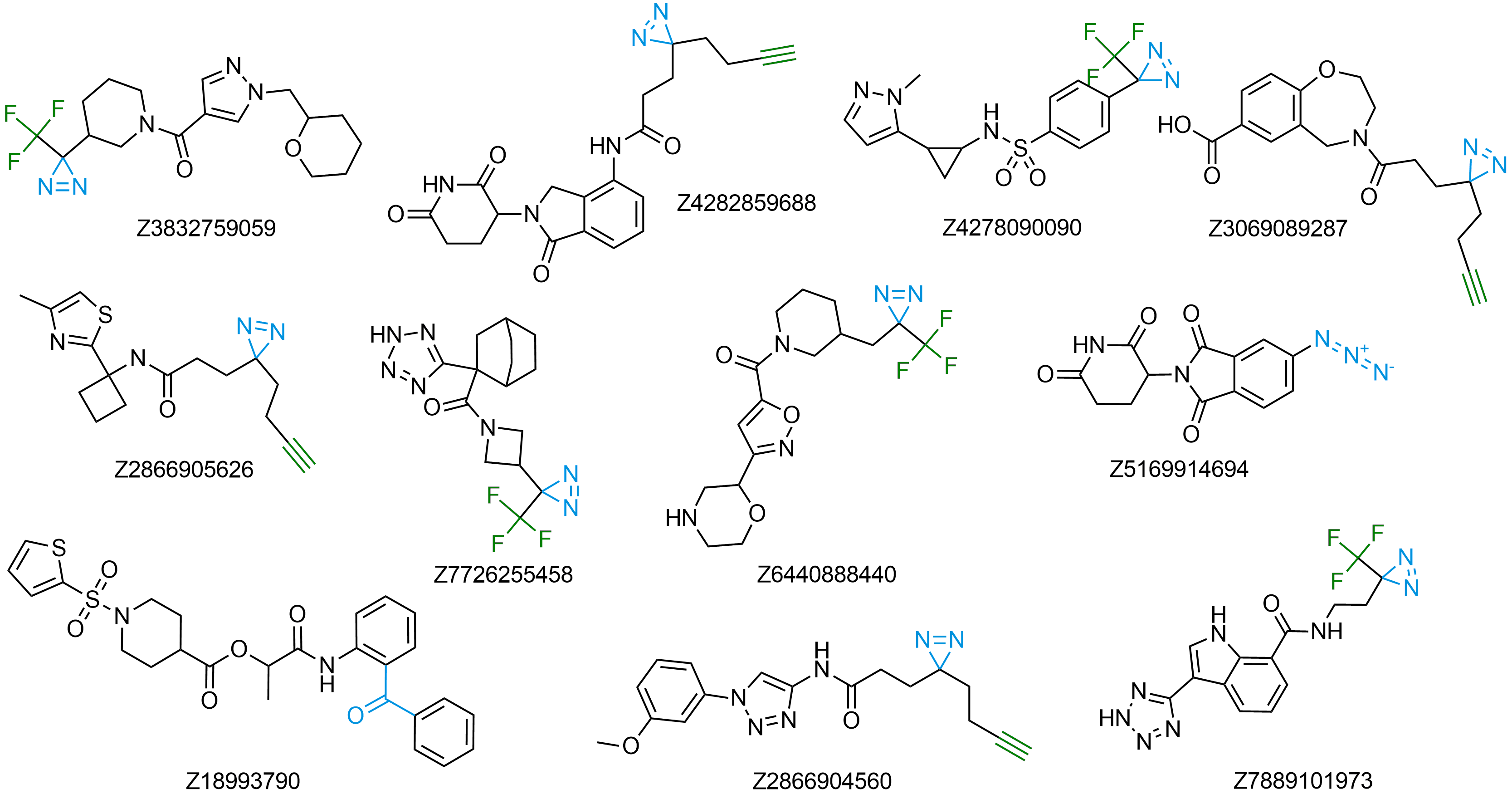
Selected publications
-
A direct-to-biology high-throughput chemistry approach to reactive fragment screening.
Thomas R. P. et al. Chem. Sci. 2021, 12, 12098-12106. DOI: 10.1039/D1SC03551G -
Ligand and Target Discovery by Fragment-Based Screening in Human Cells.
Parker C. G. et al. Cell. 2017, 168 (3), 527-541. DOI: 10.1016/j.cell.2016.12.029 -
Fully Functionalized Small-Molecule Probes for Integrated Phenotypic Screening and Target Identification.
Cisar J. S. and Cravatt B. F. J. Am. Chem. Soc. 2012, 134, 25, 10385–10388. DOI: 10.1021/ja304213w
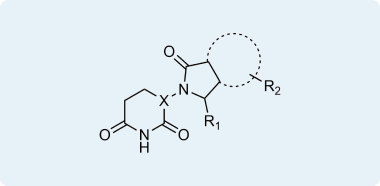
CRBN binders and their functionalized analogs
Cereblon (CRBN) is one of the most explored E3 ligases extensively used to construct PROTACs, search for new Molecular Glues, and develop functionalized tool compounds. The most extensive research has been done in this area. CRBN ligands are also known to have a good ADME profile, allowing for the development of orally bioavailable PROTACs.
We have been working with Thalidomide-like and Glutarimide chemistry for years and gained versatile experience in this chemistry field. Our experienced chemists are continuously working on the design and synthesis of new attractive Building Blocks, while our parallel chemistry group extensively elaborates approaches in the modification of the most interesting intermediates. This knowledge allowed us to enumerate a REAL Array of over 18 million CRBN-focused compounds, which can be synthesized within only 4 weeks and with 80% success rate.
Screening Compounds and Libraries
Over 15 000 CRBN focused compounds are available from stock including Glutarimides, IMiD Library Uracil-like, and Avadomide derivatives. All products are constantly updated with newly synthesized compounds. Covalent modifiers are represented by diverse and the most reliable warheads focusing on Cys, Lys, and Ser residues.
Download SD files
Plated
Stock
REAL
Product catalog
IMiD Library
IMiD-4900
Size
4 900
compounds
Descriptions
Designed and specially synthesized for the discovery of Immunomodulatory Imide Drugs
Download file
CRBN-MG-4560
Size
4 560
compounds
Description
Designed to capture the widest possible range of novel chemotypes of CRBN binders, providing a foundation for the discovery of next-generation Molecular Glues
Download file
PAG-640
Size
640
compounds
Description
Built upon novel Phenyl Amino Glutarimide (PAG) analogs, these compounds combine structural innovation with potent activity — offering a fresh perspective in Molecular Glue design
Download file
PD-880
Size
880
compounds
Description
Chemical stability, cellular potency, and permeability are just a few of the advantages that make Phenyl Dihydrouracil (PD)-based CRBN ligands promising candidates for optimal Molecular Glue discovery
Download file
PG-800
Size
800
compounds
Description
Phenyl Glutarimide (PG)-based library was purposefully developed to expand the structural diversity space of Molecular Glues
Download file
Len Library
Len-640
Size
640
compounds
Description
Lenalidomide- and Pomalidomide-scaffold based — a time-tested backbone, reimagined for modern Molecular Glue design
Download file
AAG Library
AAG-1600
Size
1 600
compounds
Description
Acylated Amino Glutarimide (AAG) featuring 5- and 6-membered heterocyclic systems: a promising platform for the discovery of next-generation Molecular Glues
Download file
Avadomide library
AVD-560
Size
560
compounds
Descriptions
A special selection of new Avadomide analogs
Download file
Diverse CRBN Library
CRBN-960
Size
960
compounds
Descriptions
Designed to cover all structural diversity of CRBN binders, a key component of the E3 ubiquitin ligase complex
Download file
CRBN Covalent Library
CCRBN-160
Size
160
compounds
Descriptions
A small selection of diverse covalent binders capable of interacting with CRBN
Download file
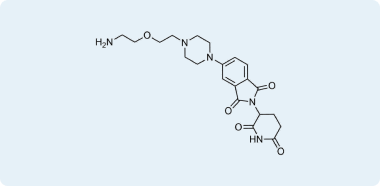
Representative examples of compounds in pre-plated CRBN Diversity Library

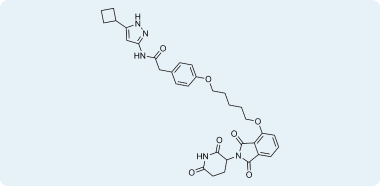
Examples of compounds in pre-plated CRBN Covalent Library

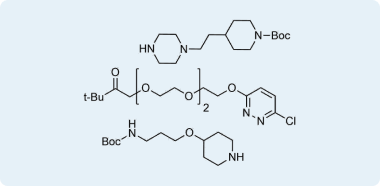
Examples of in-stock CRBN binders with linkers

Latest Arrivals
Glutarimide ring variation
Imide motif variation
Core structure variation
Designed to target DCAF family of E3 ligases
5 440 pre-plated compounds
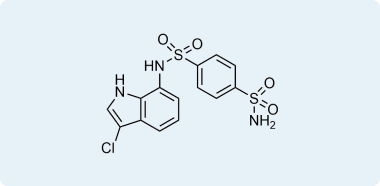
For a long time, Indisulam and a few other clinically tested aryl sulfonamides such as Tasisulam and chloroquinoxaline sulfonamide (CQS) remained compounds with pronounced selective in vitro anticancer activity and unclear action mechanisms. Recent extensive investigations in cancer genome sequencing together with X-Ray crystallography and cryo-EM showed that these compounds act as a “molecular glue” to induce degradation of RBM39 via complex formation and recruiting CUL-4-DCAF15 E3 ligase. This insight provided structural and mechanistic data that significantly extended our understanding of their mode of action. Further research in this area reveals covalent binders of other members of DCAF family proteins, such as DCAF1, DCAF11, and DCAF16.
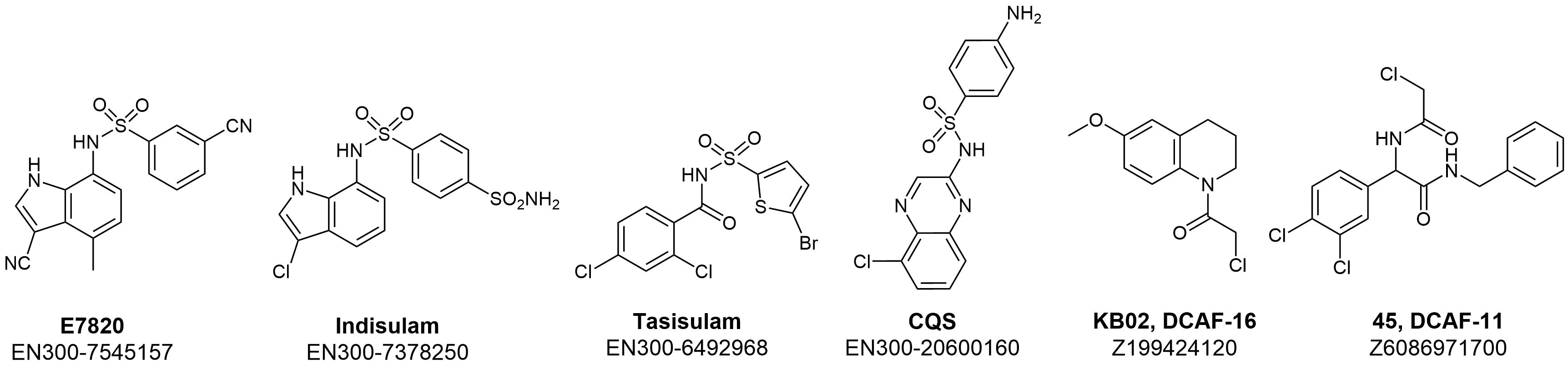
Recent structural data and reported DCAF binders allowed us to design a dedicated library of compounds with the potential to act as molecular glues. The library is now available in a pre-plated format and can be quickly delivered in any convenient for your project format.
Download SD file
Typical Formats
Catalog No.
DCAF Library
DCAF-5440-10-Y-10
Compounds
5 440
17 plates
Amount
10 µL of 10 mM DMSO solutions
Plates and formats
384-well Echo plates, Labcyte #LP-0200, 320 compounds per plate, first two and last two columns empty
Price
Catalog No.
DCAF Library
DCAF-5440-50-X-20
Compounds
5 440
68 plates
Amount
50 µL of 20 mM DMSO solutions
Plates and formats
96-well plates, 80 compounds per plate, first and last columns empty; Greiner #781270
Price
*We will be happy to provide our library in any other most convenient for your project format. Please select among the following our standard microplates: Greiner Bio-One 781270, 784201, 781280, 651201 or Echo Qualified 001-12782 (LP-0200), 001-14555 (PP-0200), 001-6969 (LP-0400), C52621 or send your preferred labware. Compounds pooling can be provided upon request.
Library Design
DCAF ligases have become more and more promising targets in protein degradation drug discovery. Proven efficacy and convenient chemistry make these targets attractive for developing new probes and drug candidates rapidly.
According to the structural data Indisulam and Tasisulam bind DCAF15 in an overall configuration similar to E7820, maintaining the backbone hydrogen bonds from the sulfonyl groups to DCAF15 Ala234 and Phe235 and the water-mediated hydrogen bonds. However, the methyl-to-hydrogen substitution at C4 in Indisulam limits the hydrophobic interactions with DCAF15 Val477 and Val556, while Tasisulam lacks the indole NH hydrogen bond to the backbone carbonyl of DCAF15 Phe231.
Based on the available structural data we have designed a library of potential SPLAMs from our stock Screening Collection using the following approach:
- Scaffold-based selection of potential DCAF binders using a set of known binders for DCAF15, DCAF16, DCAF11, and DCAF1.
- Generation and selection of close chemotypes/scaffolds based on diverse aryl and heteroaryl (five-membered inclusive) sulfonamides;
- Further selection based on pharmacophore modeling and docking results using available structural data of DCAF15;
- MedChem filtering according to criteria of lead/drug-likeness.
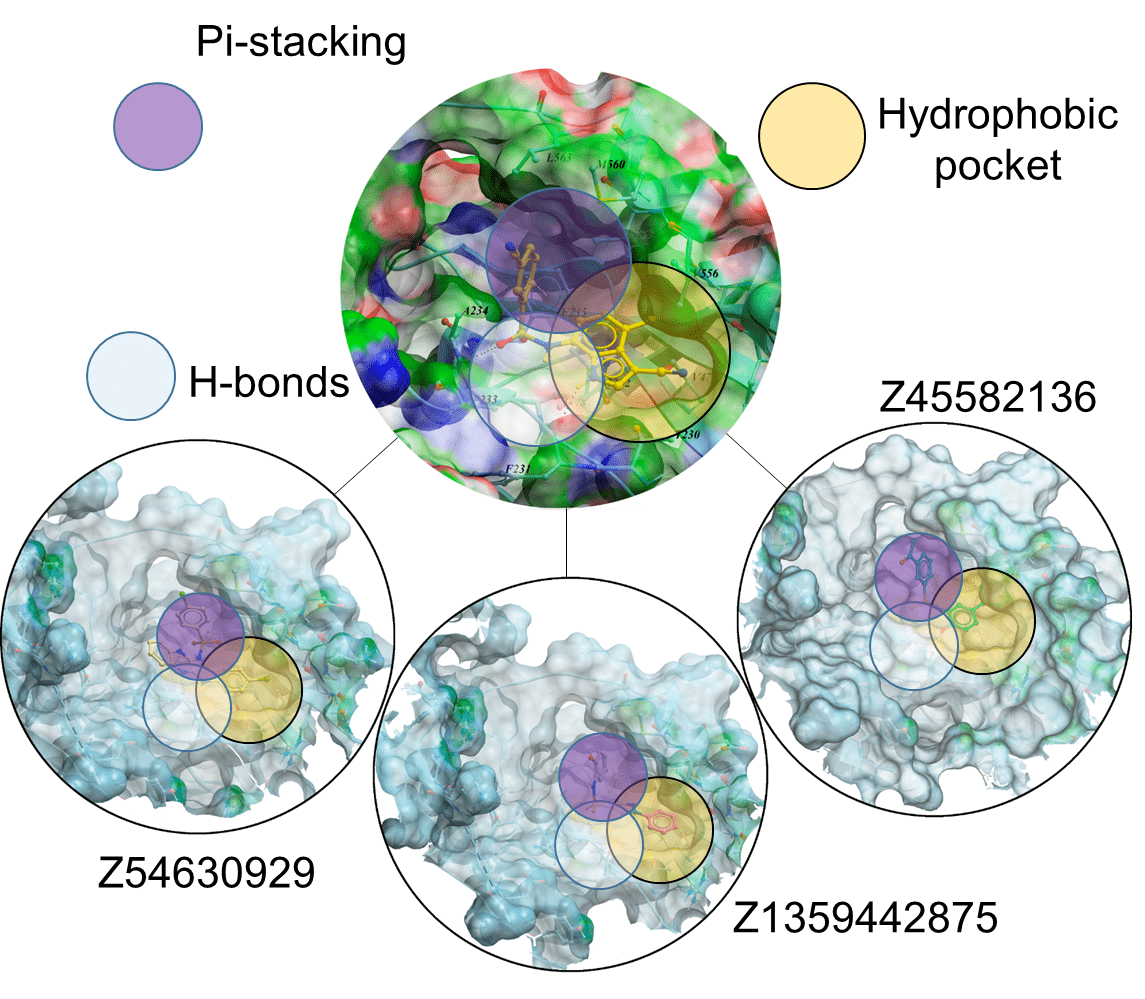
Docking results
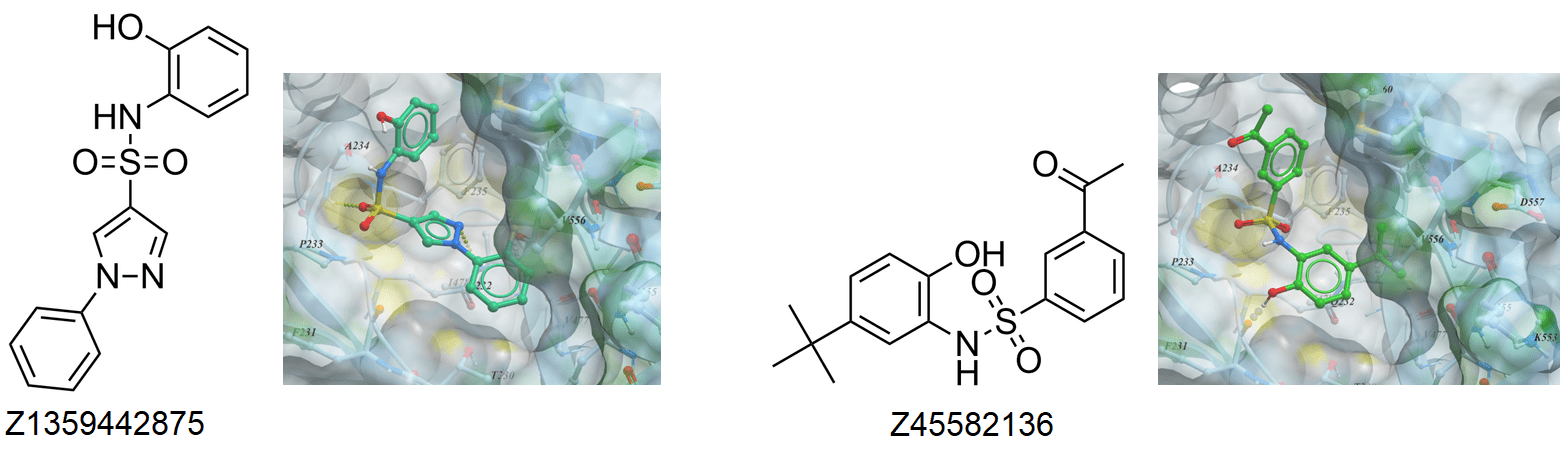
Examples of SPLAM analogues
Ready-to-use tools for your discovery
The recently emerged therapeutic area that modulates the ubiquitin-proteasome system needs new and reliable chemistry. Booming biological assays in this field of drug discovery require the production of hundreds and thousands of new functional intermediates, bifunctional molecules, and new molecular glues. The time needed to produce new derivatives is crucial at any stage of the drug discovery program. We offer advanced synthetic chemistry support, including custom library design and synthesis.