6 119 compounds in stock and over 1.1 million in REAL
960 compounds pre-plated Sulfonyl Fluoride Fragment Library
640 compounds pre-plated Sulfonyl Fluoride Screening Library
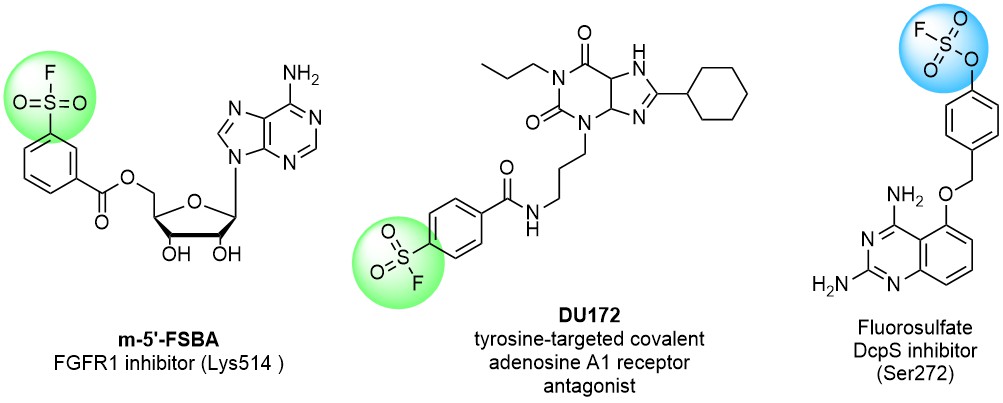
Sulfonyl fluorides are becoming increasingly popular in covalent probe discovery and are often used as tool compounds in chemical biology. Their high stability and low reactivity made them promising in modern drug discovery. Sulfonyl fluorides are known to react with multiple nucleophilic amino acid residues including Tyr, Lys, His, Arg, Ser and Thr. That is especially important due to the much higher abundance of Ser and Lys in proteins over Cys (on average about 9% and 6% vs 2%).
Download SD files
Plated libraries in stock:
There is a significant difference in reactivity between fluoro sulfonates, sulfamoyl fluorides, and alkyl aryl/hetaryl sulfonyl fluorides, which enables the design of selective covalent binders. For example, less reactive fluoro sulfonates have been reported to react better with Tyr phenolic hydroxy groups than sulfonyl fluorides and sulfamoyl fluorides.
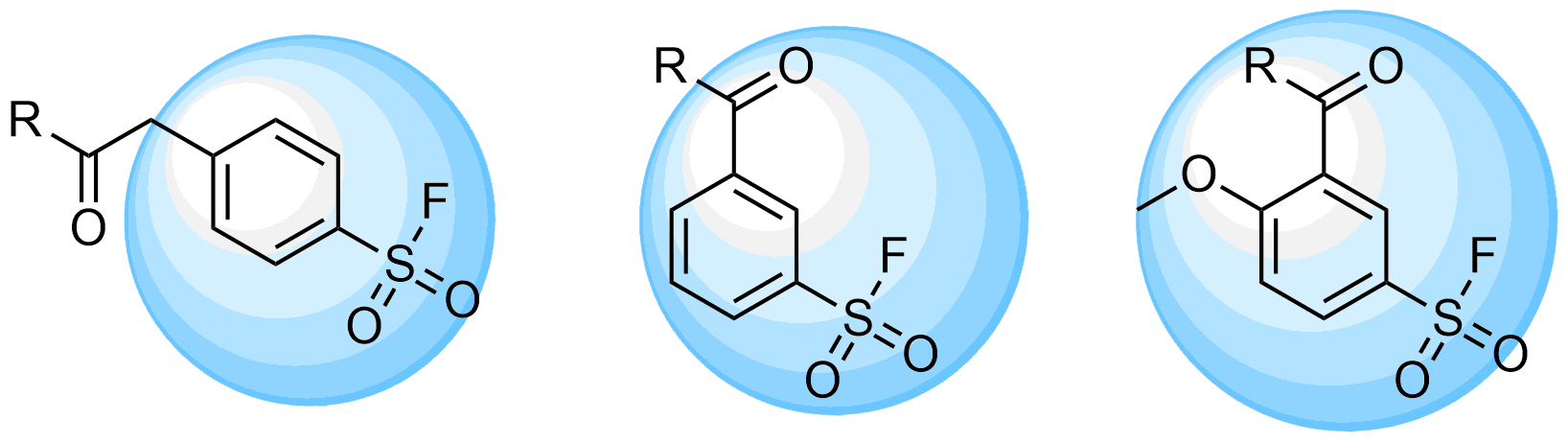
A recently published study by the research group at GSK on hydrolytic stability and reactivity of various sulfonyl fluorides, reveals interesting findings and a way to profile sulfonyl fluorides. Several substituted benzene sulfonyl fluoride scaffolds have been reported to possess balanced reactivity due to enough stability to hydrolysis and moderate activity toward nucleophiles.
REAL Sulfonyl Fluorides
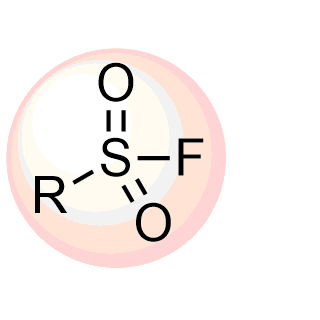
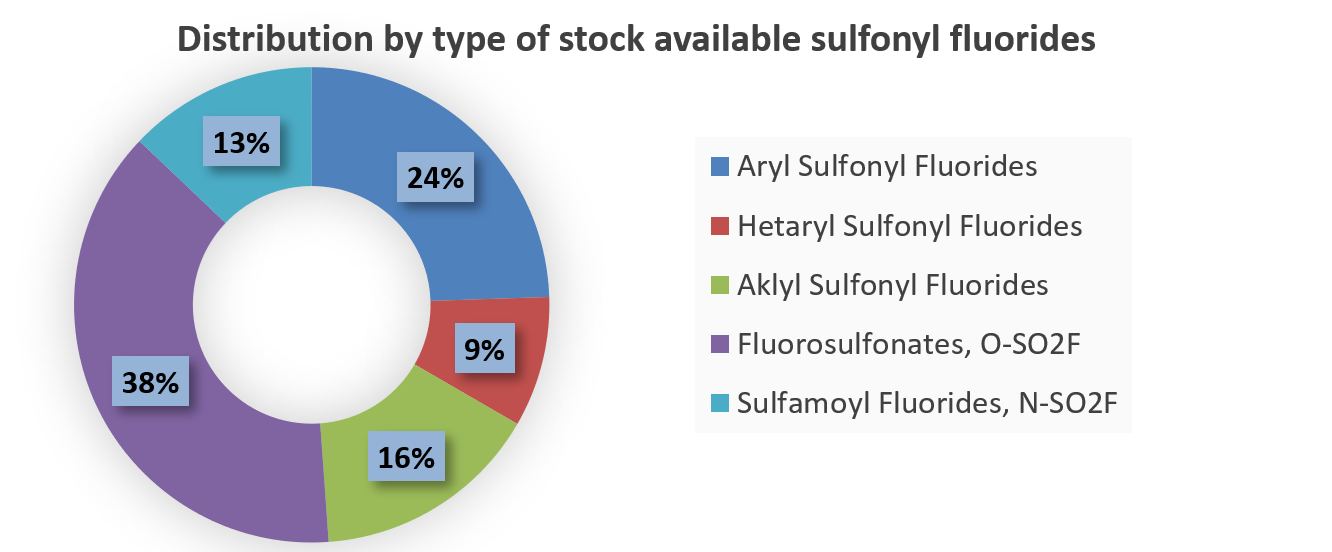
Over 701k REAL Sulfonyl fluorides, including aryl, hetaryl and alkyl sulfonyl fluorides
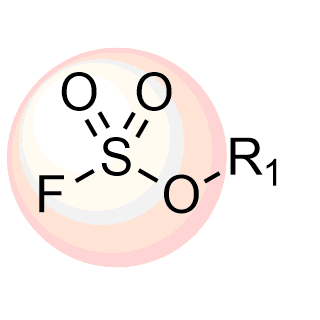
Attractive REAL Fluorosulfonates, over 94k compounds available for fast parallel synthesis with at least 80% success rate
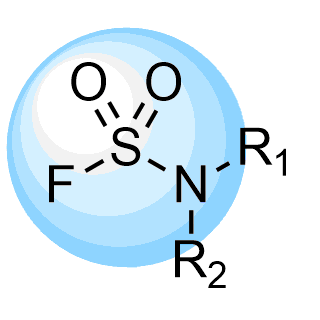
REAL Sulfamoyl fluorides, 210k for fast parallel synthesis within 3 weeks only
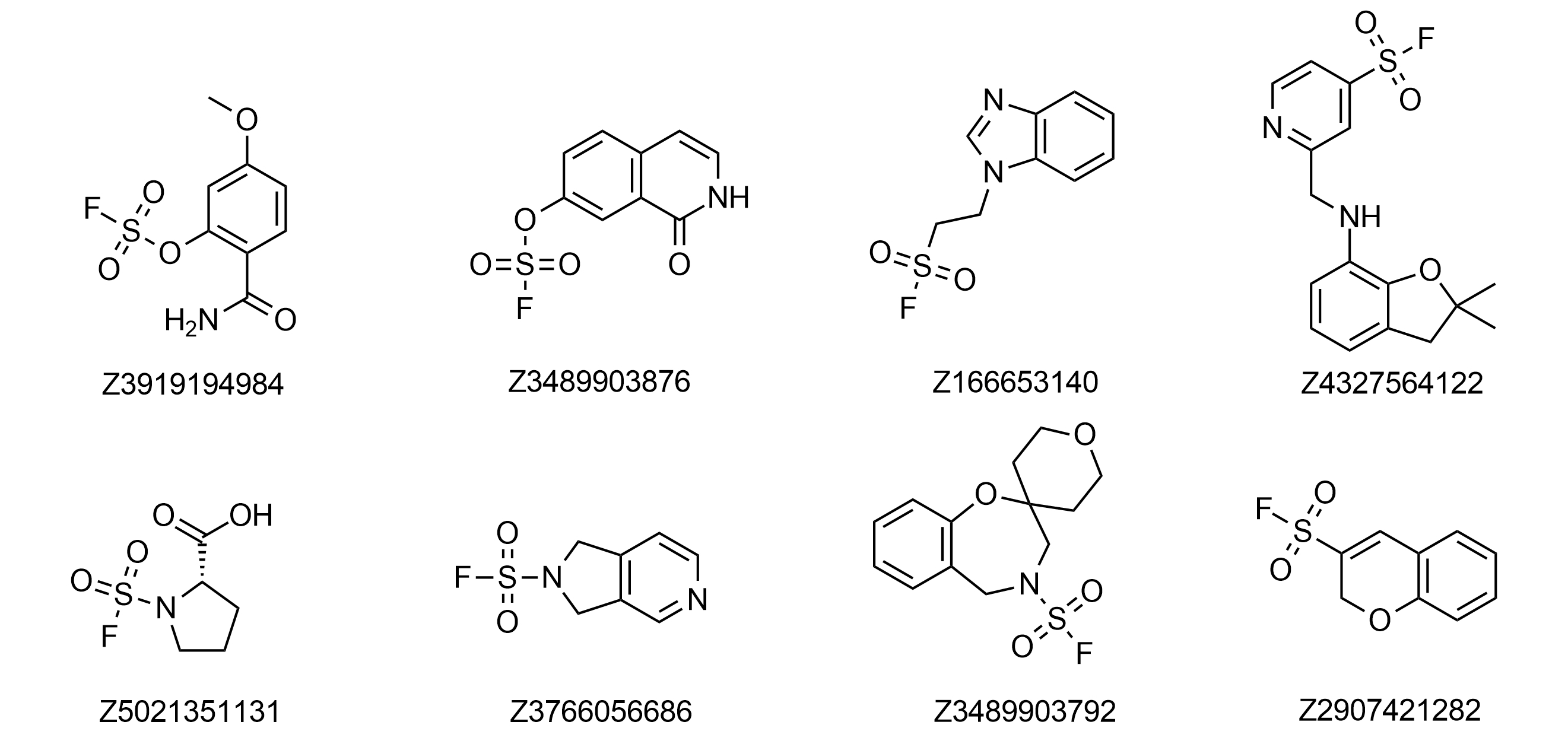
Examples of pre-plated Sulfonyl Fluorides
Covalent Screening Library: 640 Sulfonyl Fluorides plated at 10 mM in DMSO
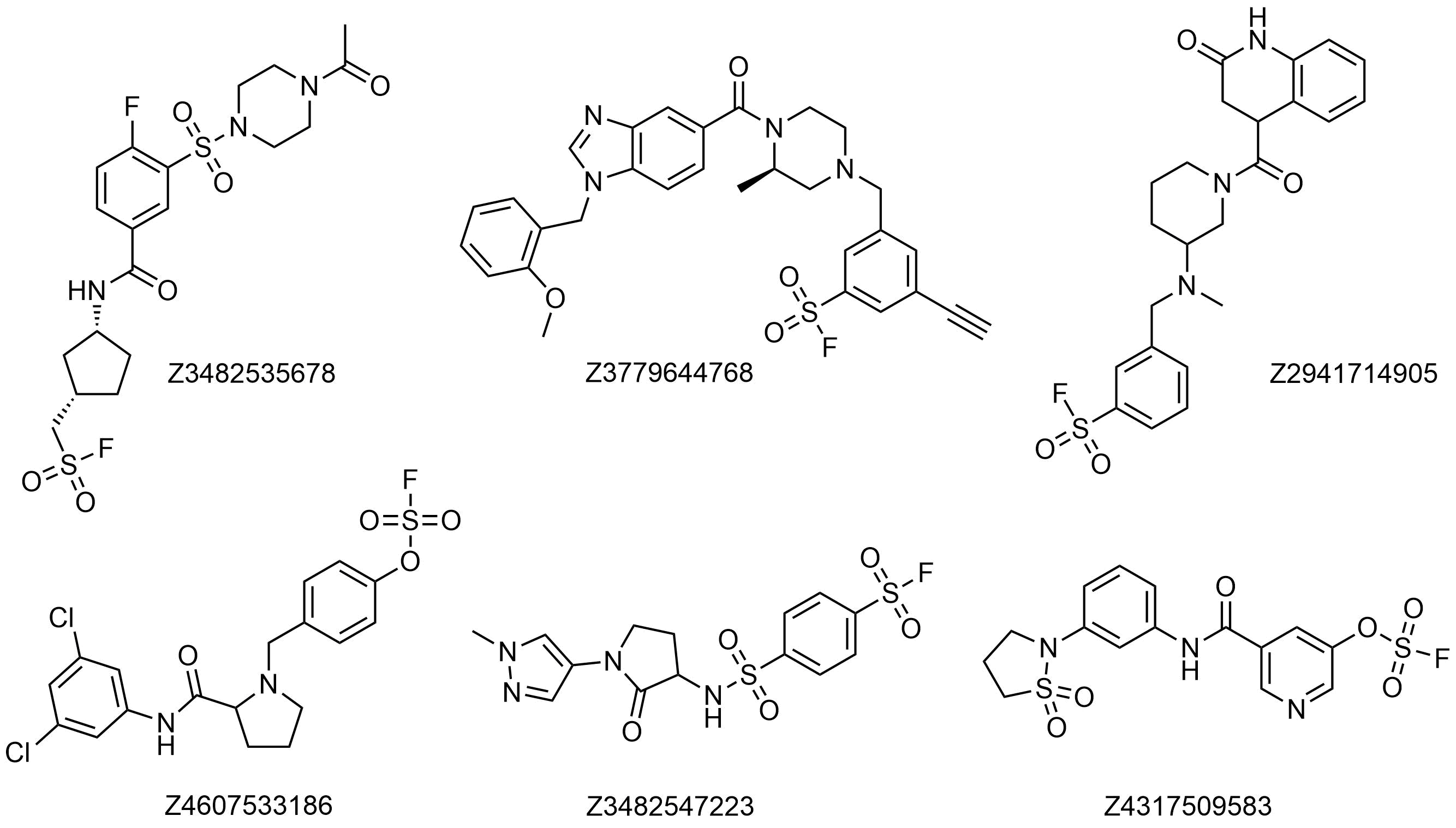
Covalent Fragment Library: 960 Sulfonyl Fluorides plated at 100 mM