Promising and more stable isosteres of acrylamides
555 compounds in stock 320 pre-plated compounds
Bicyclo[1.1.0]butane carboxylic amides emerged as a new class of Cys-focused electrophiles. They are considered as bioisosters of acrylamides, but their increased stability and unique reactivity can result in the development of new selective covalent binders.
We offer over 500 compounds from stock and continuously work on the synthesis of new derivatives. You can select from our growing REAL Array of currently 1 million strained bicyclobutanes and be the first to try them on your target. Synthesis of hundreds of compounds will take us a few weeks only.
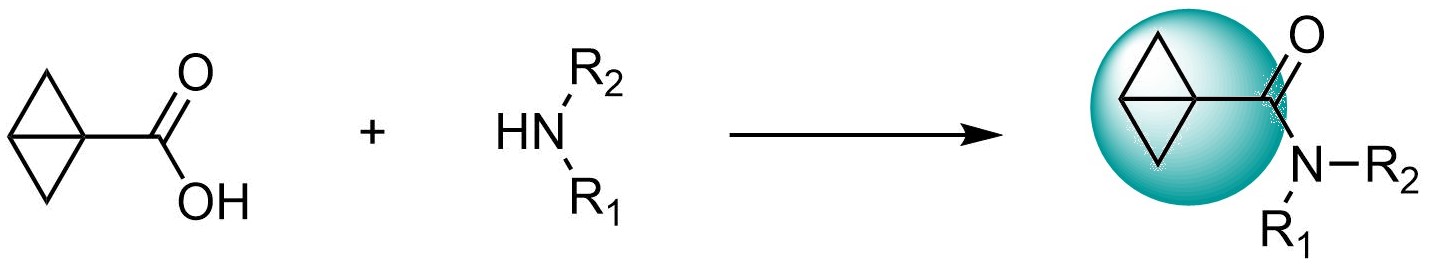
New Bioisosteres of Acrylamides readily available through custom parallel synthesis and from our REAL Array
Download SD files
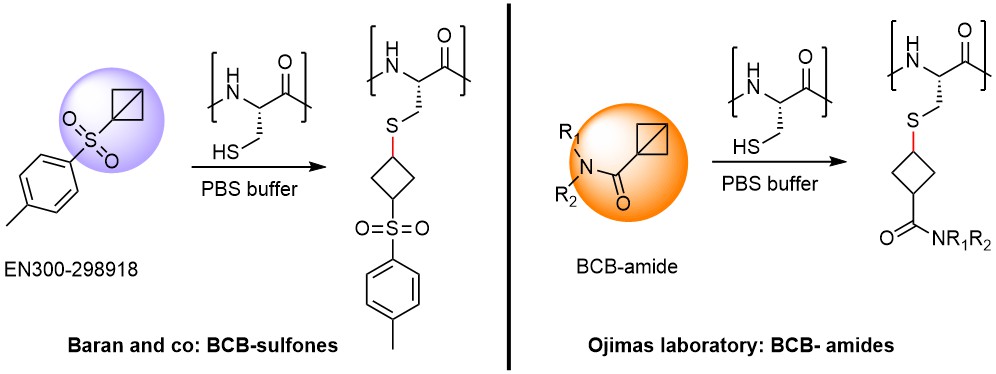
The chemistry of bicyclo[1.1.0]butane (BCB) derivatives is vividly developing. Strained hydrocarbons (e.g., EN300-298918) were proposed by Baran as suitable candidates for selective covalent labeling of Cys-residues in polypeptides. In his study, the reactivity of bicyclo[1.1.0]butyl aryl sulfones was similar to that of acrylamides in GSH-assay. Recently, Ojida introduced BCB-amide as a covalent warhead, targeting cysteine residue. These molecules are easy to synthesize from BCB-carboxylic acid and diverse amines. Importantly, BCB-amides were tested in Ramos cells, which shows their compatibility with biological systems.
BCBs represent a novel perspective area for developing new selective covalent inhibitors. We are continuously working on the enumeration and synthesis of new strained bicyclobutane derivatives.
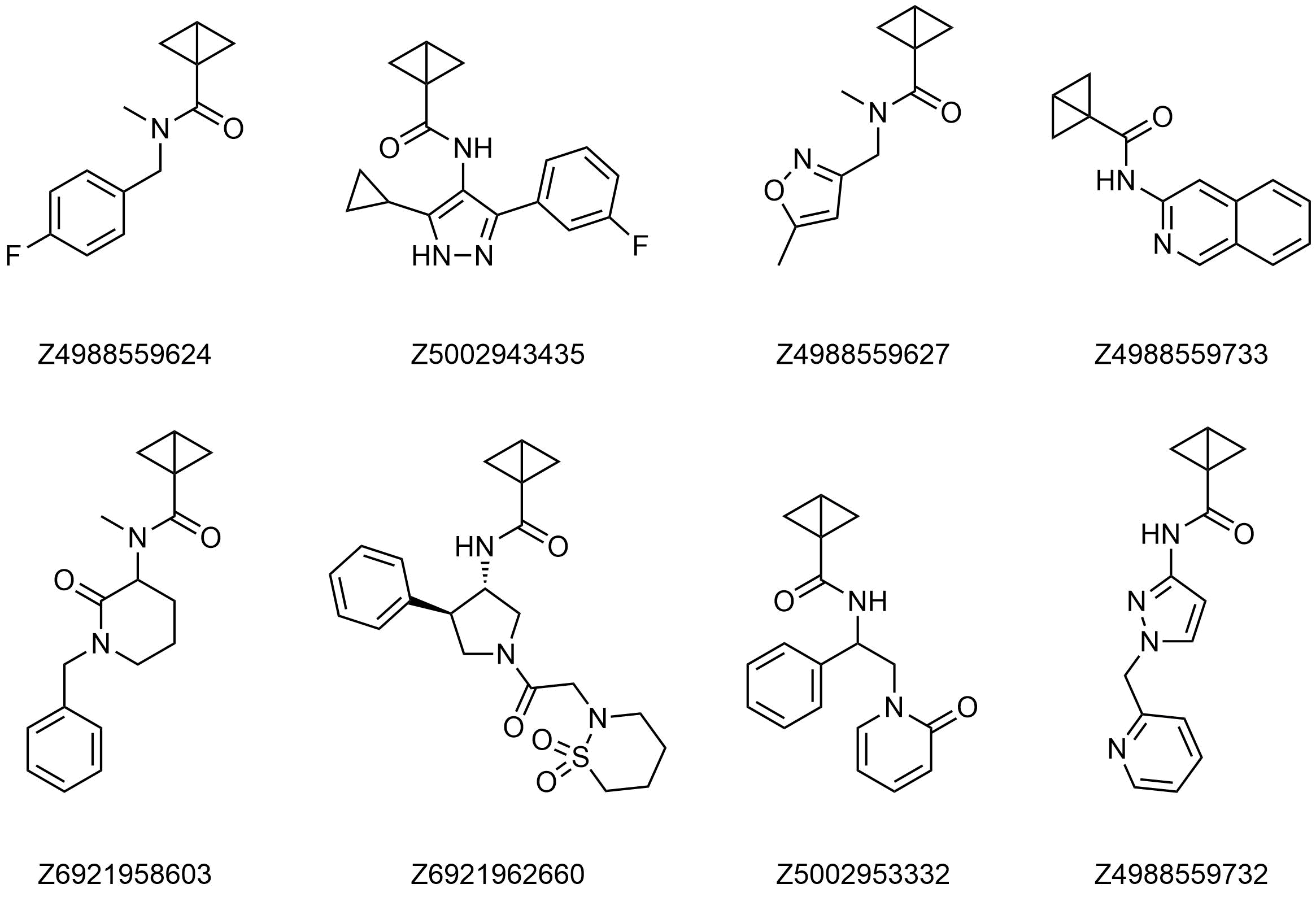
Examples of Bicyclobutane amides in stock