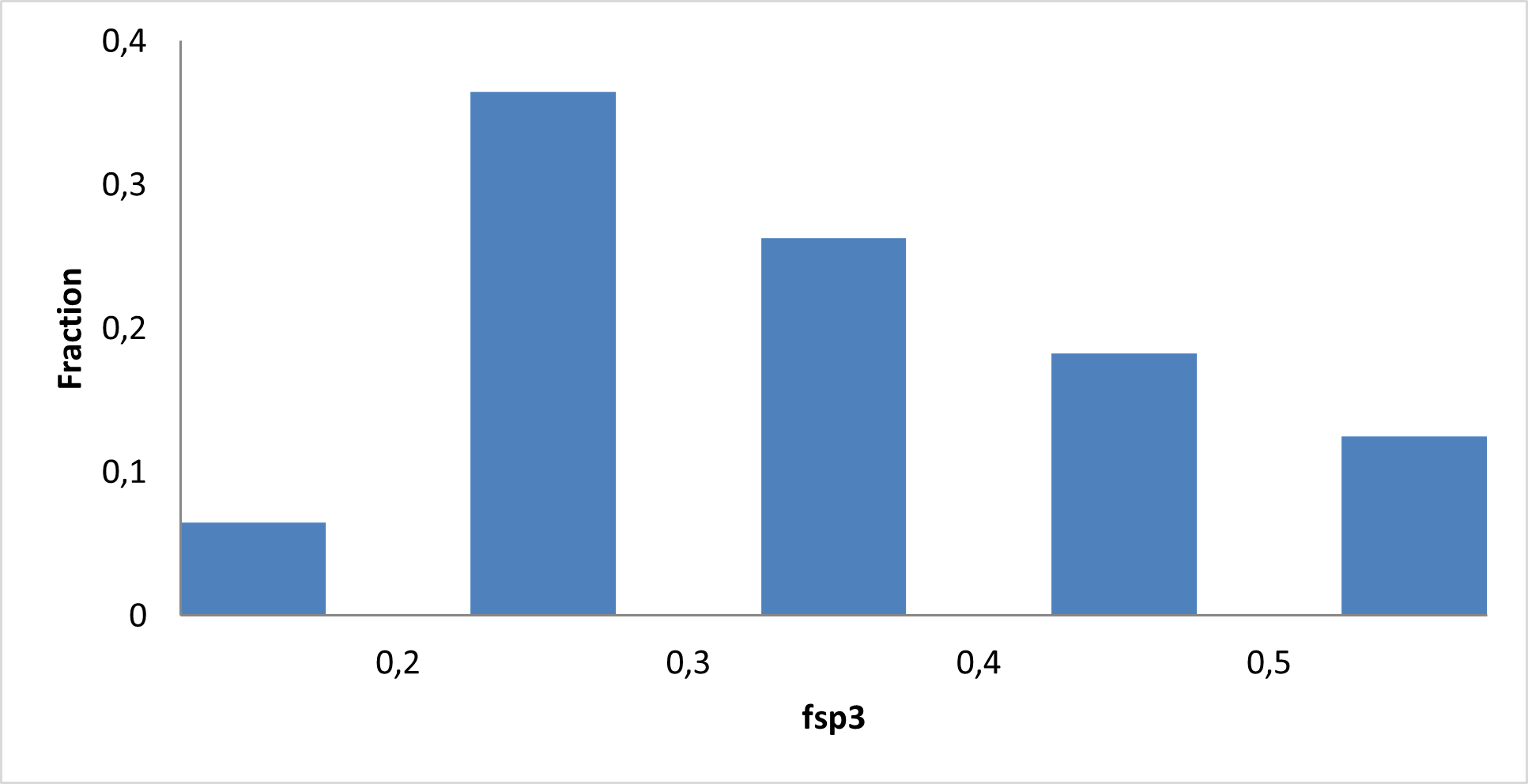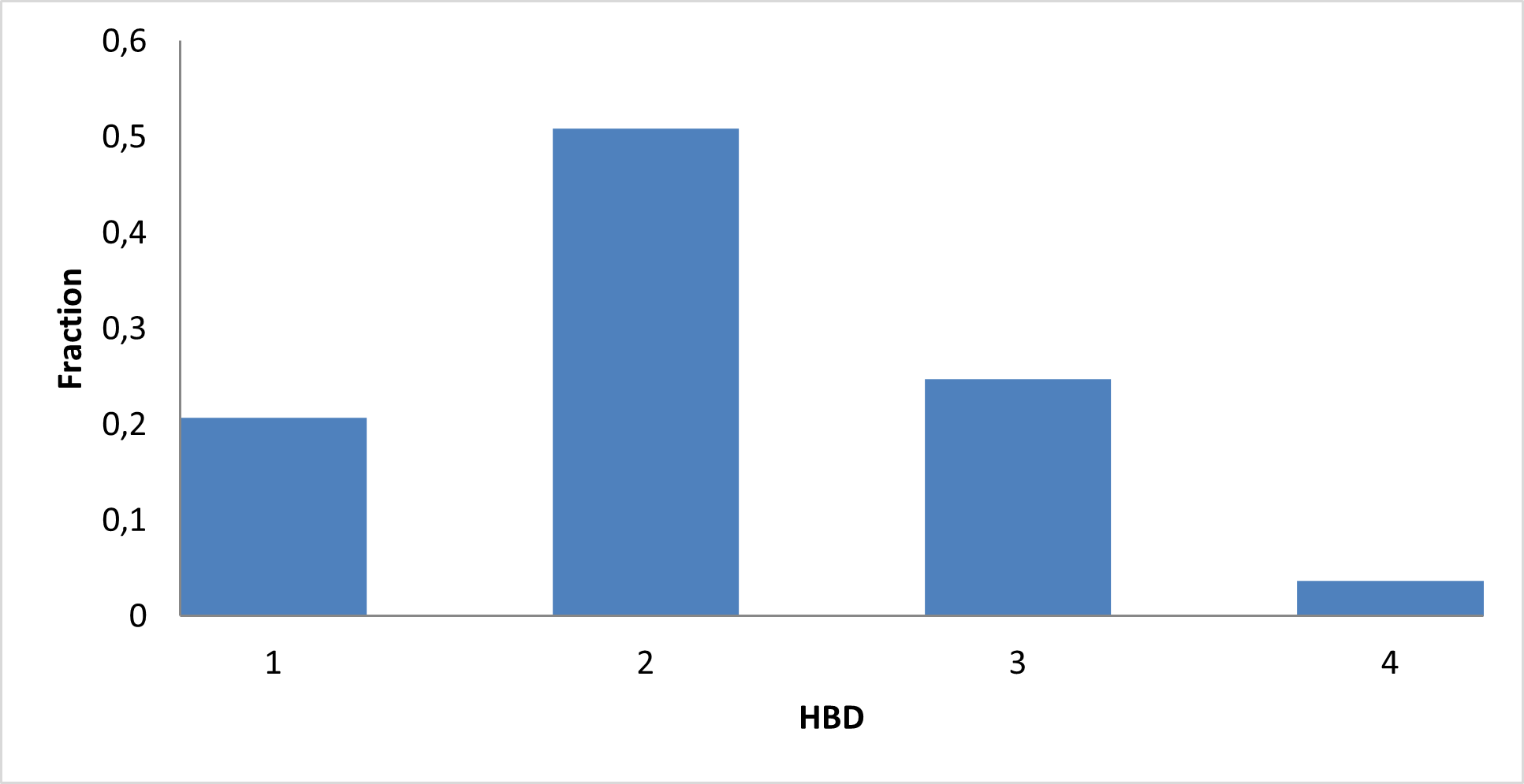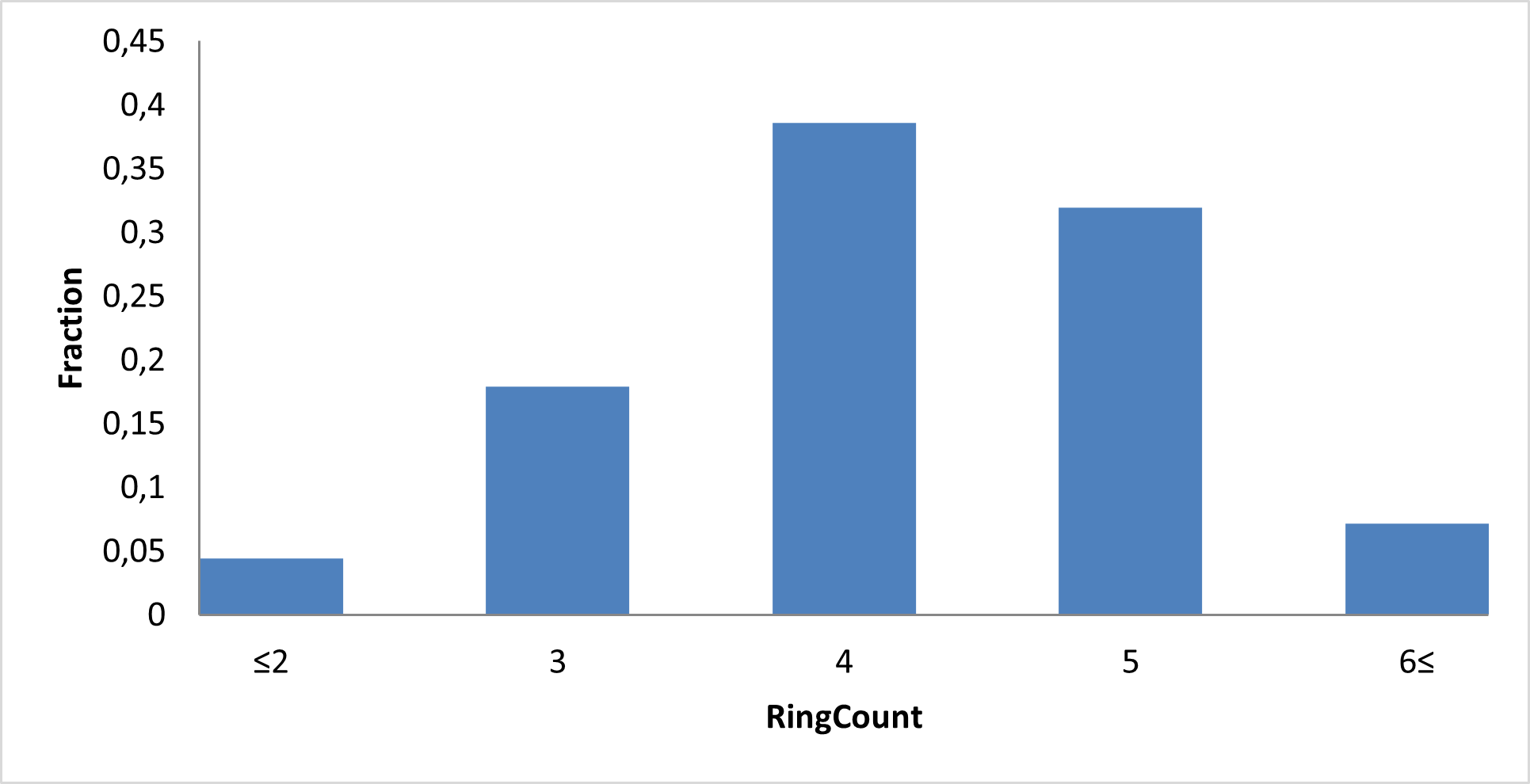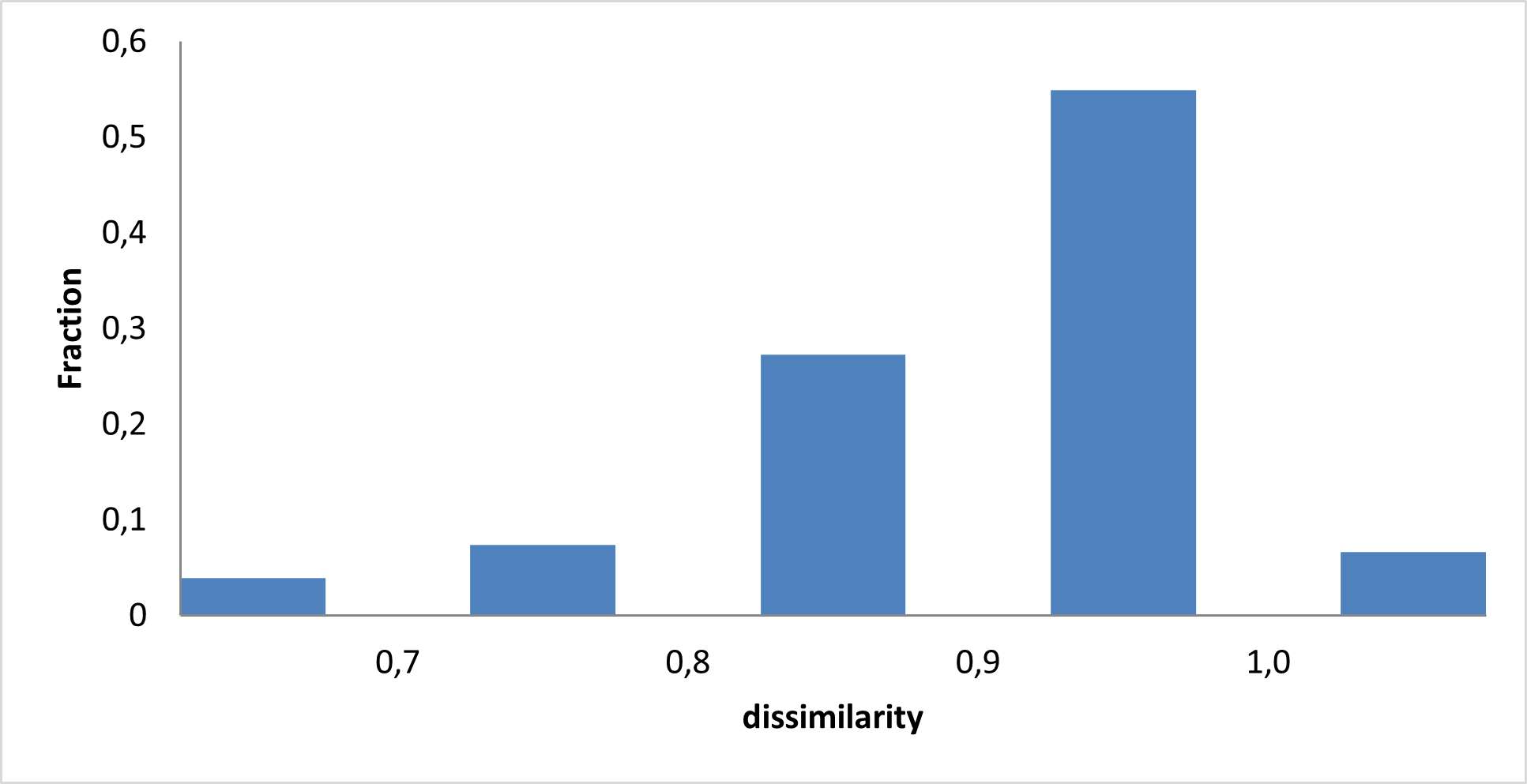A curated collection of verified CRBN binders
4 900 compounds
The IMiD Library represents our first edition of a comprehensive CELMoD (Cereblon E3 ligase modulatory drug) compound collection. CELMoDs are first class of clinically proven degraders small molecules offering new therapeutic opportunities across challenging disease areas.
Evolving from complex protein-targeting chimeras (PROTACs) to lead-like molecular glues (MGs), these molecules demonstrate improved PK/PD profiles and expanded applications, including use in CNS disorders. This rapidly advancing field holds enormous promise for discovering drugs with novel modalities.
All molecules in the library have been evaluated in newly developed assays for CRBN binding. Using an engineered full-length human CRBN construct expressed in E. coli — which eliminates the need for DDB1 co-expression—we established a cost-efficient high-throughput screening (HTS) platform. This assay was recently published in Cell Chemical Biology (2025, 32(2): 363), with binding affinity data and full experimental details provided in the supplementary materials.
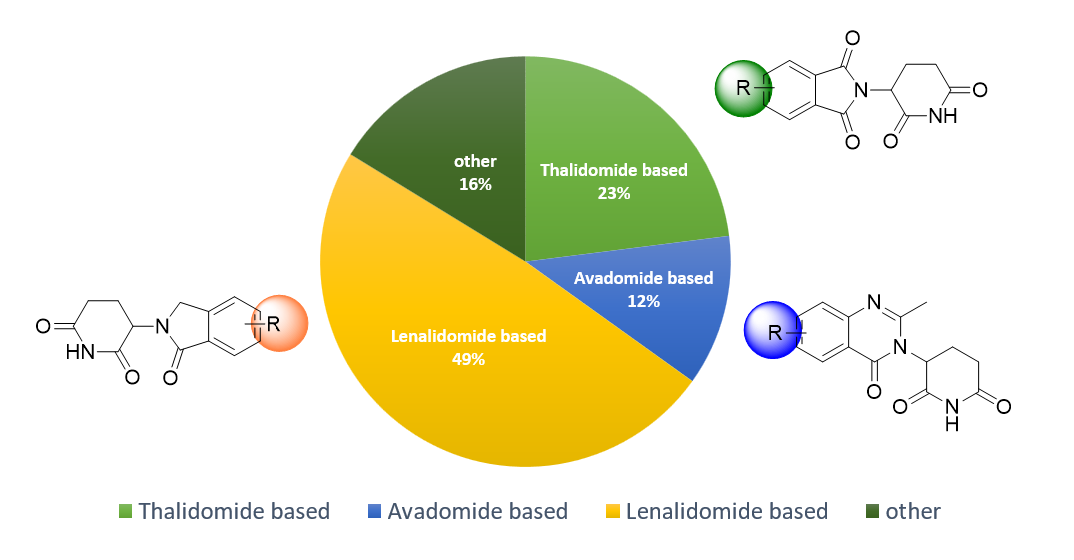
The library is primarily built around classical first-generation CRBN binders—Lenalidomide, Pomalidomide, and Avadomide— further diversified through structural modifications and variable attachment points to expand chemical diversity and functional potential.
We recommend this library as “binding first” interactome screening approach for discovery aiming on recruiting Cereblon E3 ligase.
Download SD file
Typical Formats
Catalog No.
IMiD Library
IMID-4900-10-Y-10
Compounds
4 900
14 plates
Amount
10 µL of 10 mM DMSO solutions
Plates and formats
384-well LDV Echo Qualified plates, 320 compounds per plate, first two and last two columns empty
Price
Catalog No.
Diverse CRBN Library
CRBN-960-50-Y-10
Compounds
960
3 plates
Amount
50 µL of 10 mM DMSO solutions
Plates and formats
384-well microplates, Corning #4514, 320 compounds per plate, first two and last two columns empty
Price
Catalog No.
Avadomide Library
AVD-540-25-Y-10
Compounds
540
2 plates
Amount
25 µL of 10 mM DMSO solutions
Plates and formats
384-well plates,320 compounds per plate, first and last columns empty
Price
Catalog No.
Covalent CRBN Library
CCRBN-160-20-X-20
Compounds
160
2 plates
Amount
20 µL of 20 mM DMSO solutions
Plates and formats
96-well plates, 80 compounds per plate, first and last columns empty; Grainer #781270
Price
*We will be happy to provide our library in any other most convenient for your project format. Please select among the following our standard microplates: Greiner Bio-One 781270, 784201, 781280, 651201 or Echo Qualified 001-12782 (LP-0200), 001-14555 (PP-0200), 001-6969 (LP-0400), C52621 or send your preferred labware. Compounds pooling can be provided upon request.
Library Design
Our library was designed using trusted scaffolds and enriched through a range of MedChem reliable reactions. By applying diverse synthetic strategies and carefully selected modification sites, we ensured good CRBN-binding affinity while expanding chemical diversity. The result is a structurally rich collection that offers wide chemical space coverage and serves as a powerful resource for discovery projects.
- Scaffold-based synthesis: All compounds were designed using well-established CRBN binder scaffolds with emphasis on 4- and 5-substituted Lenalidomide scaffolds, proven to tolerate modifications while maintaining strong CRBN-binding affinity.
- Synthetic diversity: Achieved through seven widely used medicinal chemistry reactions—amide coupling, sulfamidation, nucleophilic substitution, Suzuki and Buchwald couplings, urea formation, and click chemistry.
- Broad chemical space coverage: Incorporation of structurally diverse, pharmacophore-rich reagents to maximize diversity and expand chemical space.
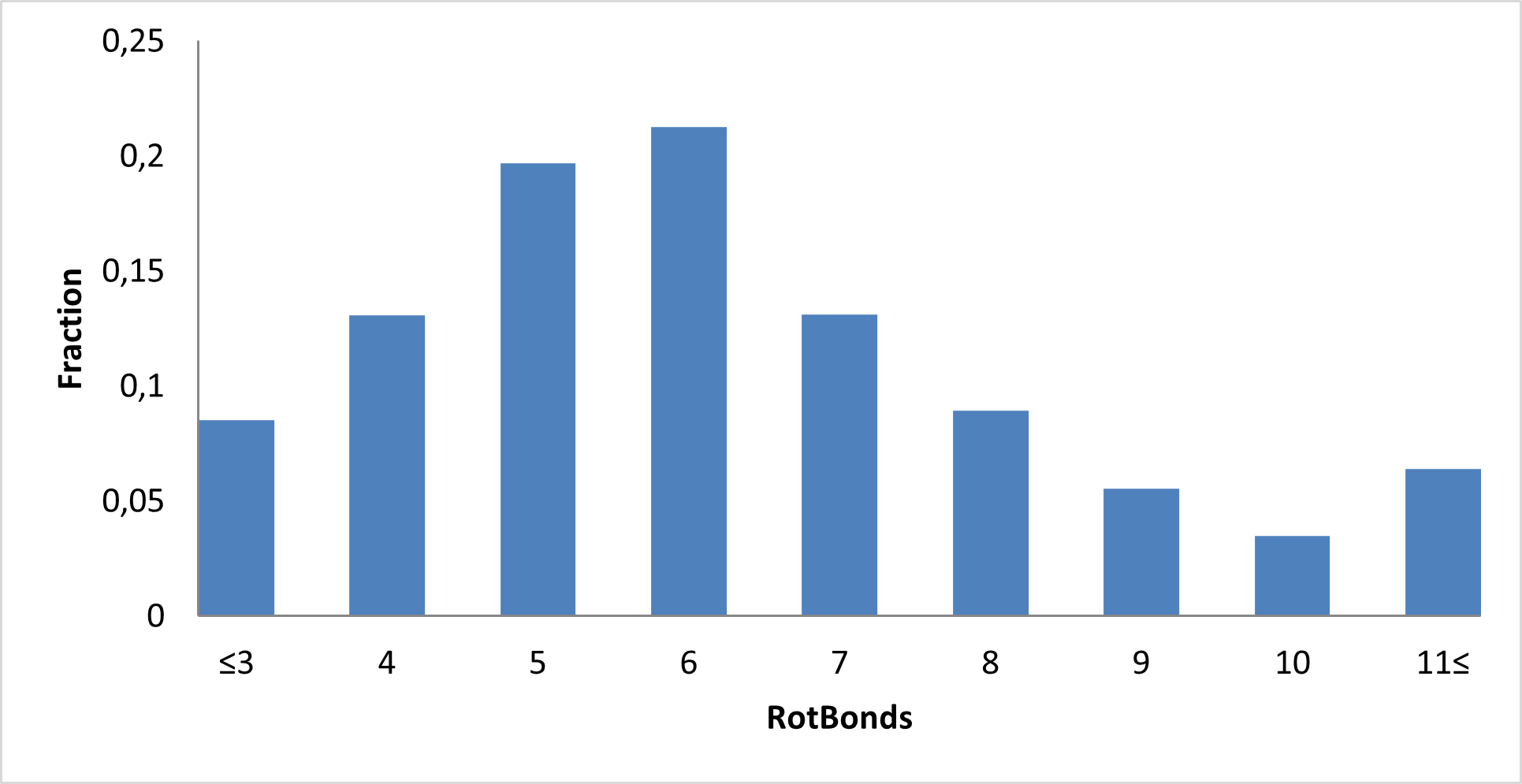
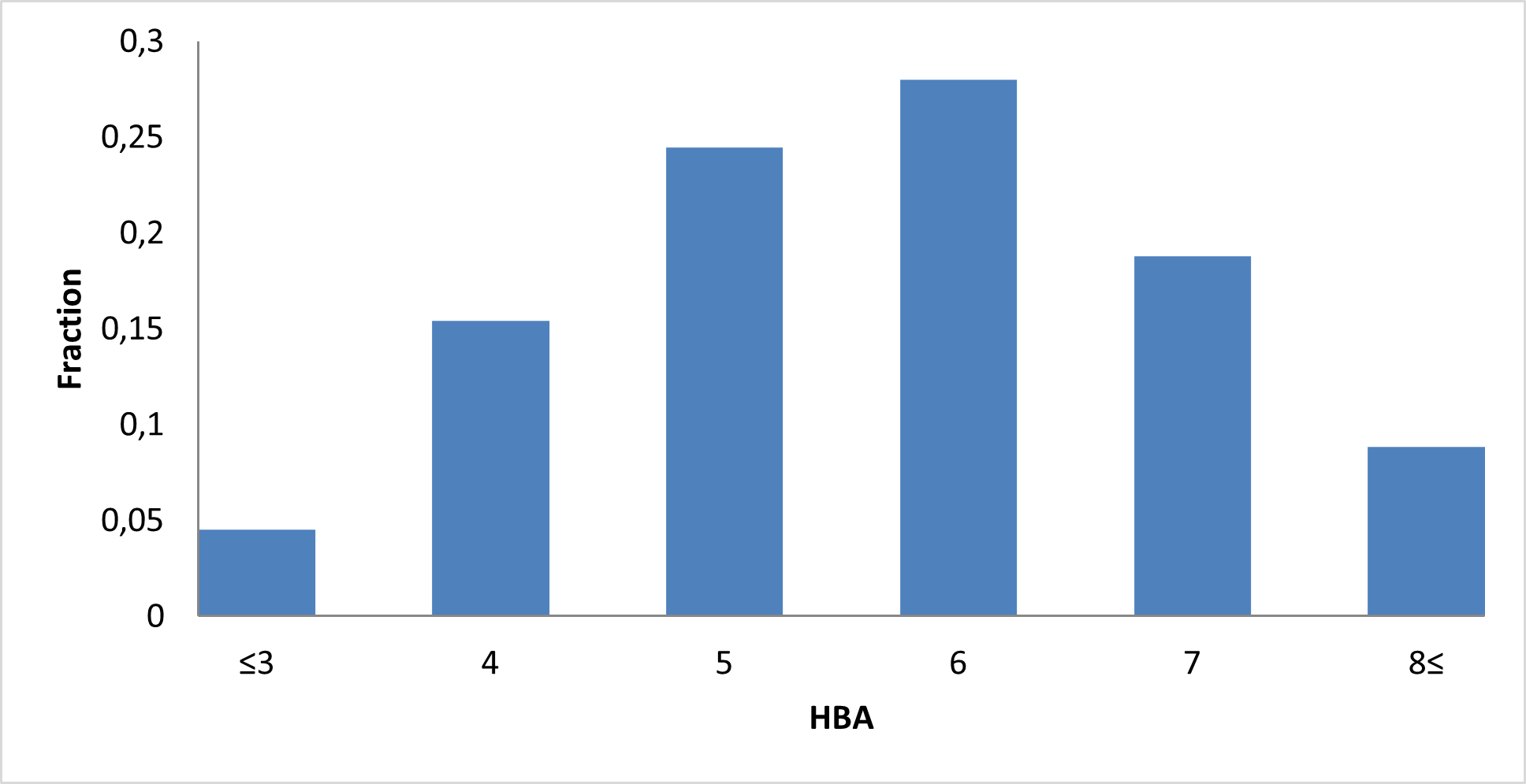
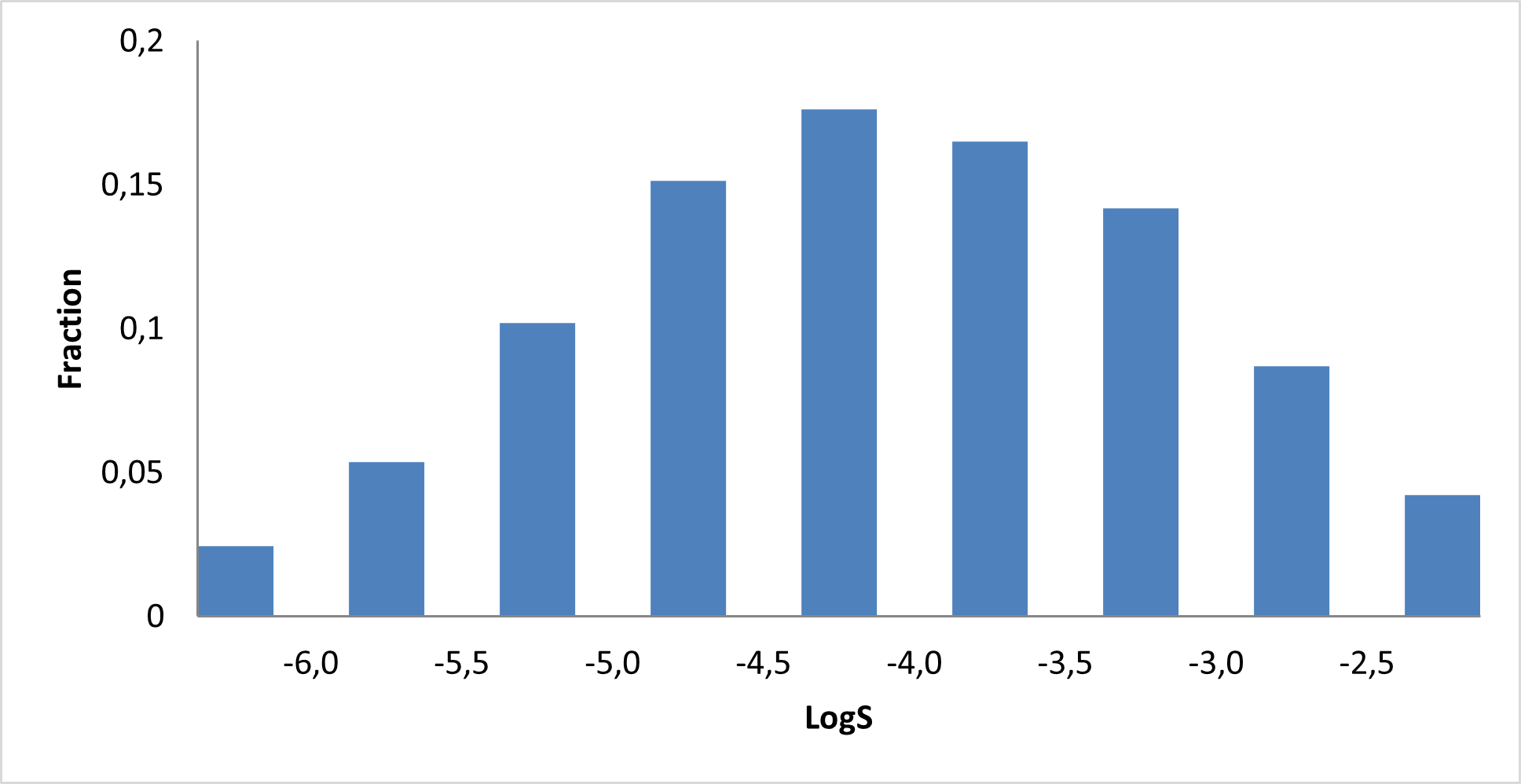
Library composition
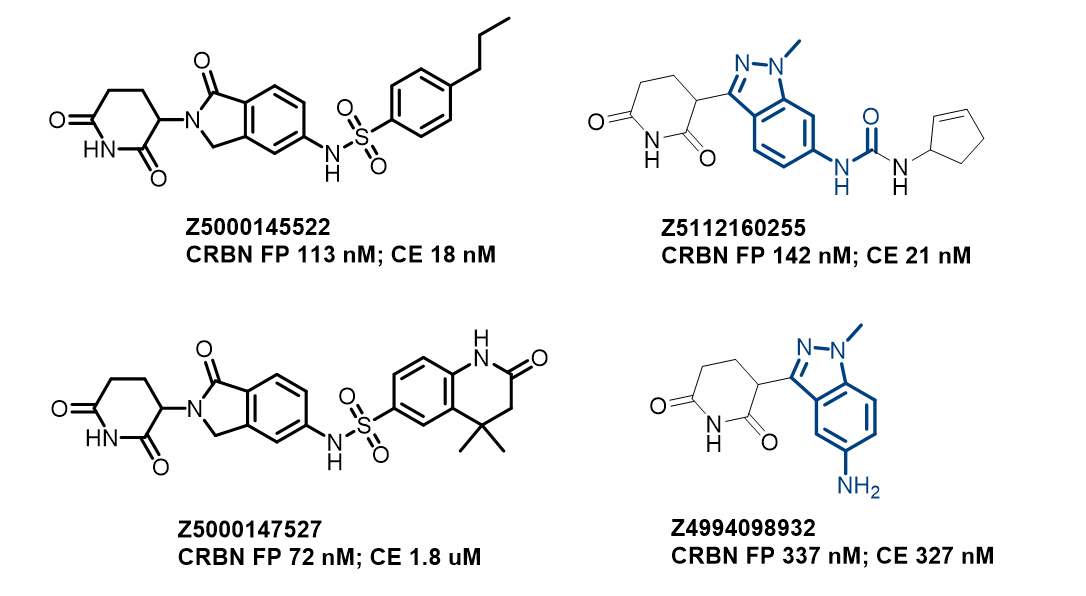
Examples of Compounds in the Library
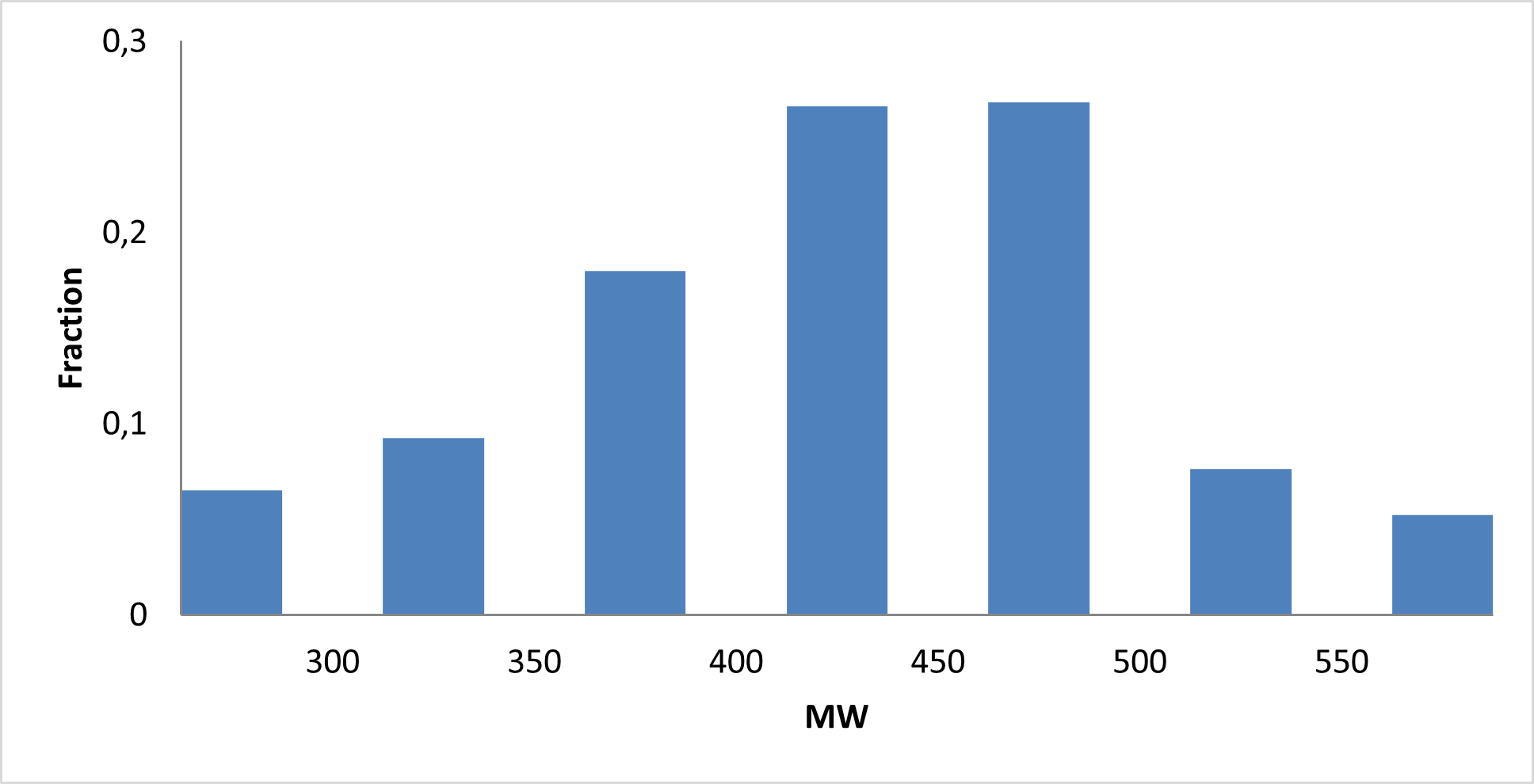
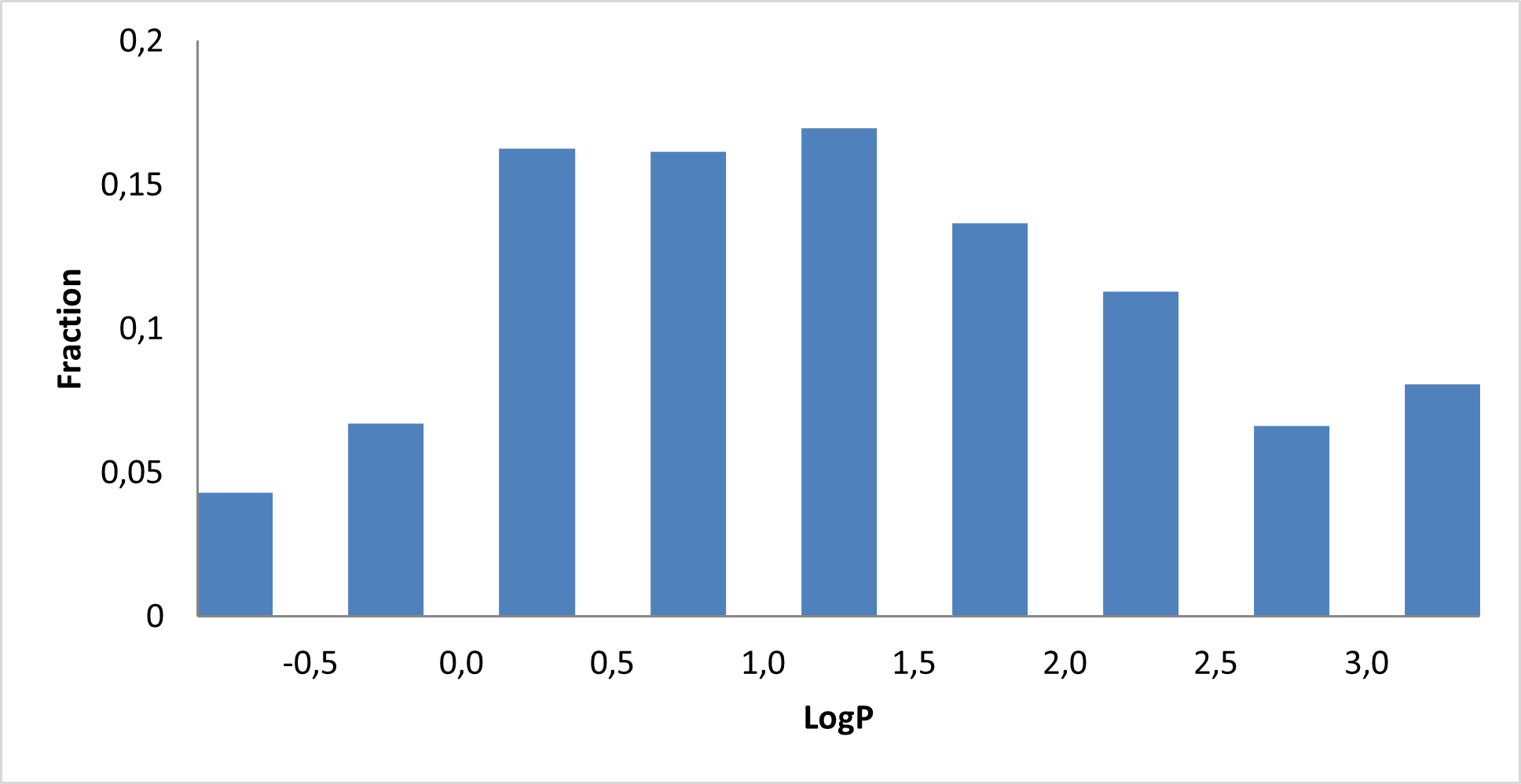
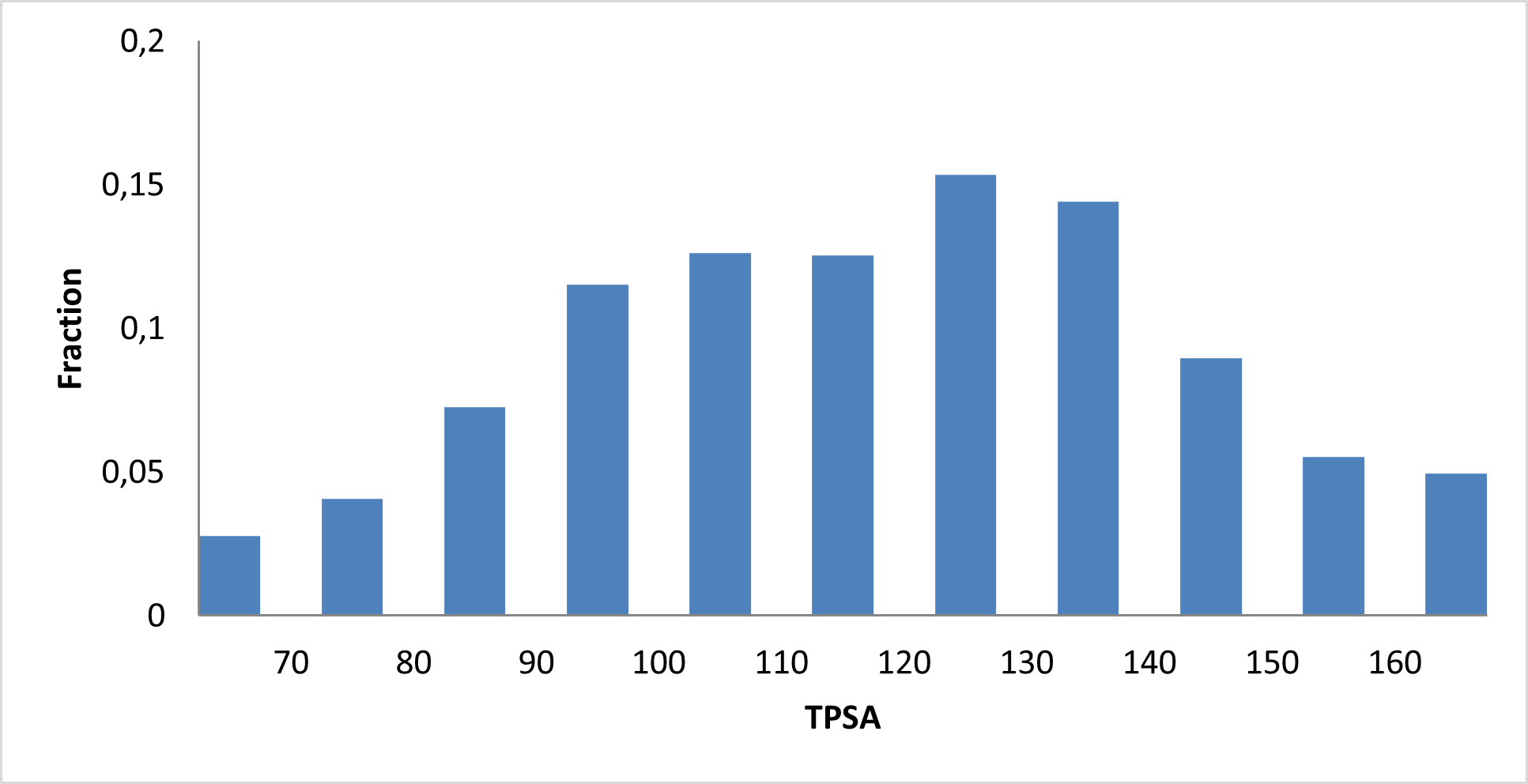
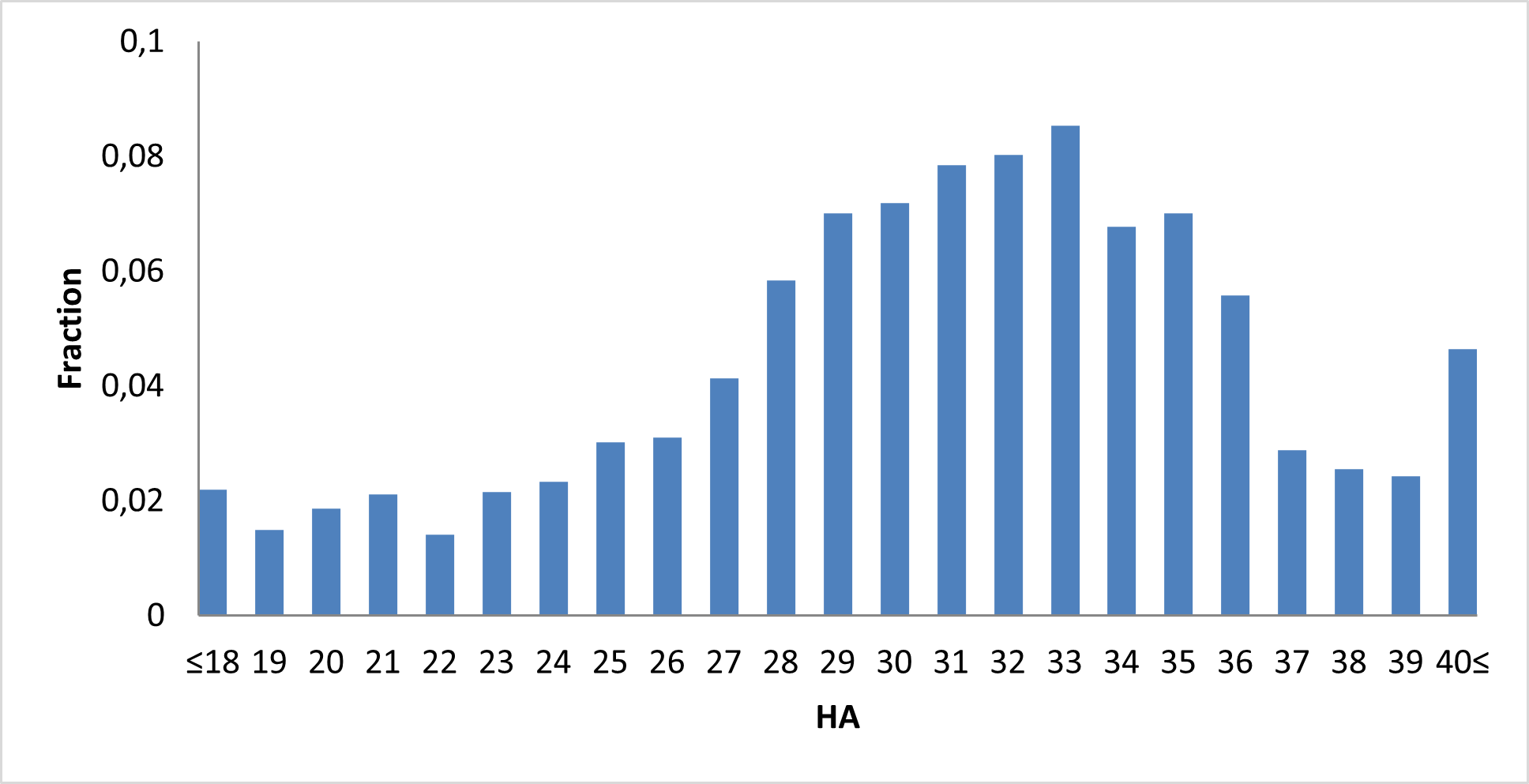
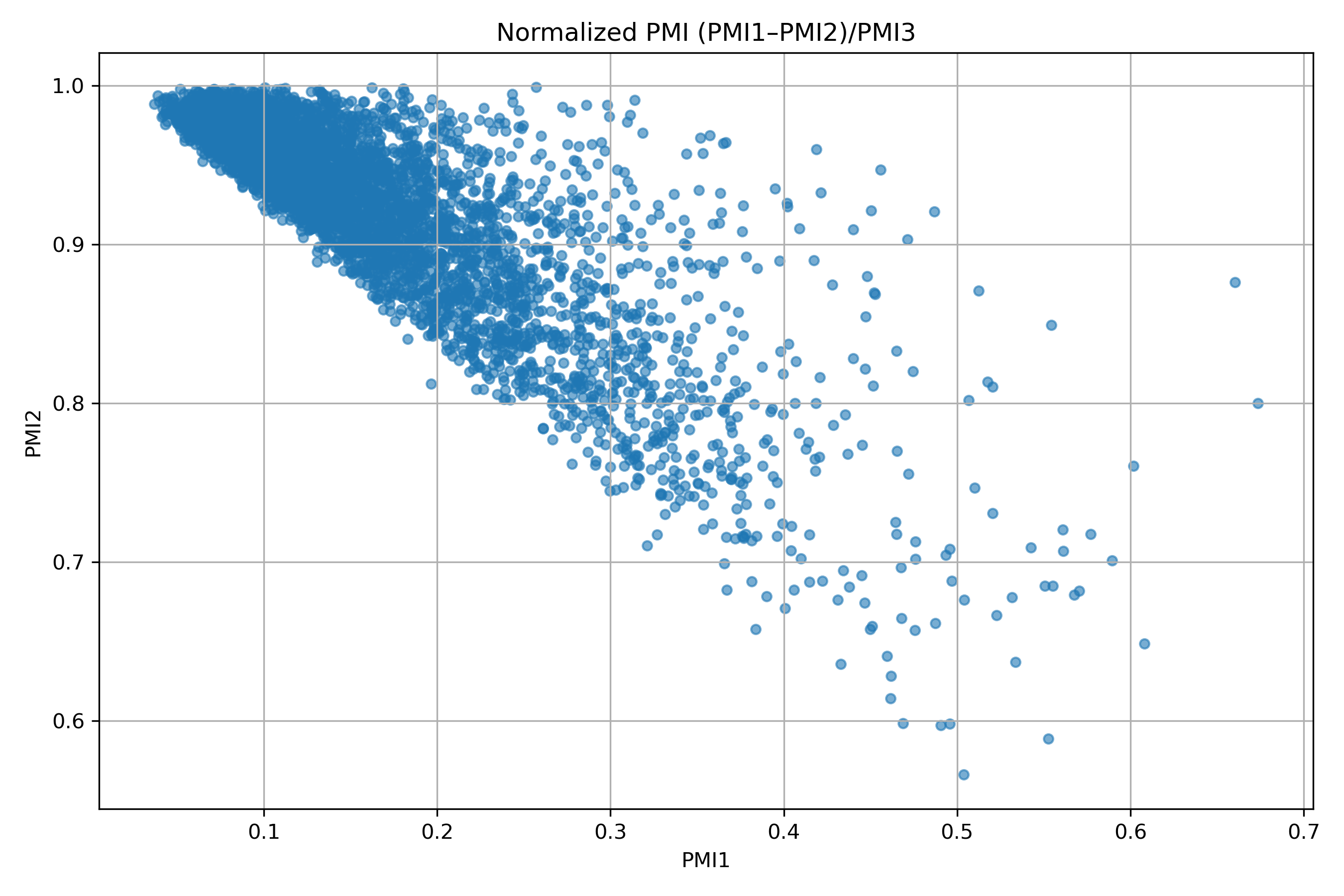
IMiD Library PMI distribution
Support
We offer comprehensive support in developing your hit compounds. Naturally such programs are realised most efficiently when biological actives originate from our screening collection. However, even if the hit compounds are from the collections of other vendors lead identification and optimization projects can proceed most productively in our hands. Sometimes for this we only need to synthesize first examples of the given chemical series and validate synthesis route.
Expanding your access to trusted pharmaceutical standards
In modern drug discovery, reliable access to well-characterized reference materials is essential for accelerating research and ensuring reproducibility.
Enamine is proud to collaborate with Pfizer to bring to the scientific community an authentic collection of reference compounds directly through our Enamine marketplace and Enamines integration capabilities (i.e. punchout site, hosted catalogs, and API solutions).
This allows researchers to explore and purchase from the catalog of selected Pfizer-provided drugs, chemical dormants, investigational compounds, and isotope-labelled molecules with appropriate quality control.
Together, Pfizer Reference Compounds and Enamine global infrastructure create a powerful resource for advancing science. Researchers can now explore new chemical space, validate findings against authentic benchmarks, and push forward the boundaries of medicinal chemistry.
Enamine offers:
- Streamline access to valuable Pfizer reference standards
- Enrich screening libraries and profiling sets
- Enhance SAR exploration with unique compounds
- Support translational and preclinical research with well-defined materials
Explore the entire catalog today and see how this collaboration can help unlock your next breakthrough.
To see more compounds in the Pfizer portfolio, visit: Enamine bioactive compounds
4 929 compounds in stock and over 19M in REAL
800 compounds pre-plated Covalent Fragments Cyanoacrylamides Library
1 920 compounds pre-plated Covalent Screening Cyanoacrylamides Library
Acrylamides are the most popular covalent warheads present in approved drugs. α-Cyanoacrylamides are a novel class of electrophilic warheads currently under active investigation in the field of covalent drug discovery. They combine moderate reactivity with high selectivity, making them an attractive alternative to irreversible warheads such as acrylamides or vinyl sulfones. Due to the presence of an electron-withdrawing cyano-group, Cyanoacrylamides are capable of forming reversible covalent bonds with nucleophilic residues, particularly the thiol groups of cysteine. This enables controlled residence time and helps to reduce the risk of off-target effects.
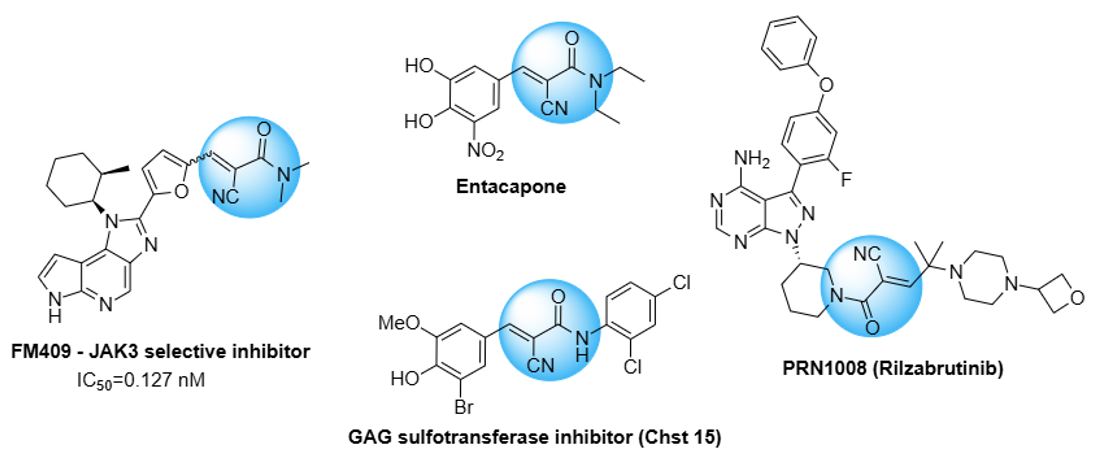
Cyanoacrylamide warheads have already been applied in a number of promising compounds, especially for kinase targeting, such as FM409 and PRN1008 (BTK inhibitor). Cyanoacrylamides can also be used for other targets, such as Chst15. An example of a related compound, Entacapone, has been approved for the treatment of Parkinson’s disease.
Enamine has almost 5k compounds in stock (90%+ pure by LCMS and/or NMR analysis), which can be delivered as dry powders or in any customized plated formats. Access to our extensive building block inventory, combined with validated synthetic approaches, enabled the creation of a 19-million-compound REAL Cyanoacrylamide Array. Compounds from this array can be synthesized within 3–4 weeks in our parallel synthesis department, with an on-time delivery rate exceeding 80%.
Explore our pre-plated Cyanoacrylamide fragments and screening libraries—ready for immediate use.
Typical Formats
Catalog No.
CACR-800-10-X-100
Compounds
800
3 plates
Amount
10 µL of 100 mM DMSO solutions
Plates and formats
384 well, Echo Qualified LDV microplates 001-12782 (LP-0200), first and last two columns empty, 320 compounds per plate
Price
Catalog No.
CSCA-1920-50-X-10
Compounds
1 920
6 plates
Amount
50 µL of 10 mM DMSO solutions
Plates and formats
384-well, Greiner Bio-One plates #781280, 1,2 and 23,24 columns empty, 320 compounds per plate
Price
*We will be happy to provide our library in any other most convenient for your project format. Please select among the following our standard microplates: Greiner Bio-One 781270, 784201, 781280, 651201 or Echo Qualified 001-12782 (LP-0200), 001-14555 (PP-0200), 001-6969 (LP-0400) or send your preferred labware. Compounds pooling can be provided upon request.
Hits derived from this library can be easily followed with analogues from stock or either synthesis of new compounds from REAL.
Download SD files
4 929 compounds for cherry-picking
Plated libraries in stock:
Library code: CSCA-1920
Version: 20 February 2023
1 920 compounds
Key features
- Reversible covalent warhead
- Cys selective
- Tuning of reactivity
Examples of the Сyanoacrylamides from stock
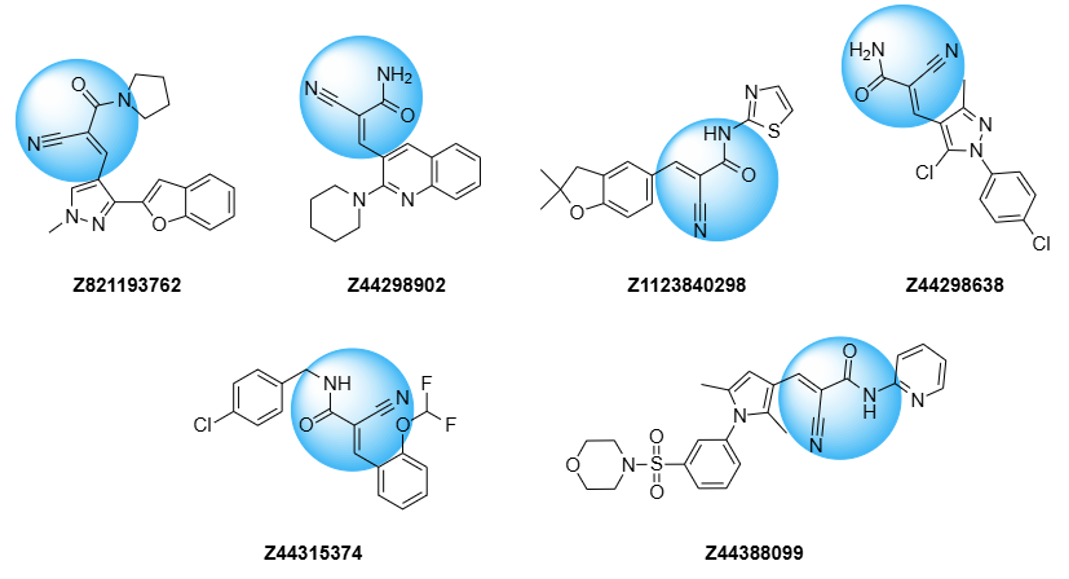
761 compounds in stock and over 920K virtual compounds
240 compounds pre-plated Covalent Screening N-nitriles Library
Cyanamides, also known as N-nitriles, have recently emerged as promising electrophilic warheads in covalent drug discovery, offering a balance between reactivity and selectivity. Their moderate electrophilicity makes them attractive alternatives to more reactive groups like acrylamides or vinyl sulfones. Cyanamides have also been explored for their ability to engage in reversible binding, forming isothioureas, which can be beneficial in certain applications.
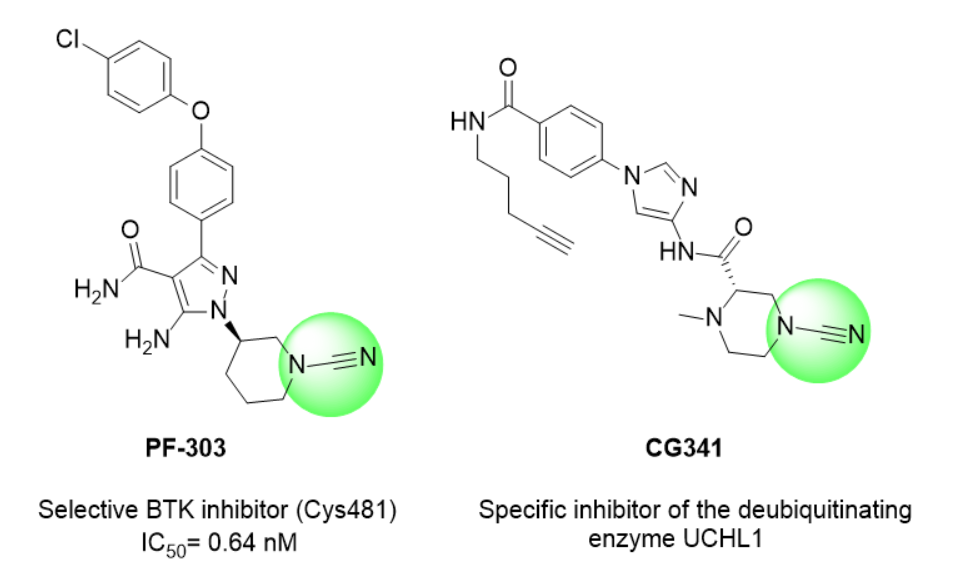
Cyanamide-based inhibitors have been applied in targeting kinases, such as BTK, and even viral proteases. Recent studies have demonstrated that cyanamides can be engineered to attain high selectivity. For instance, N-cyanopiperazines were shown to covalently and selectively inhibit the deubiquitinating enzyme UCHL1, avoiding off-target effects on closely related enzymes like PARK7.
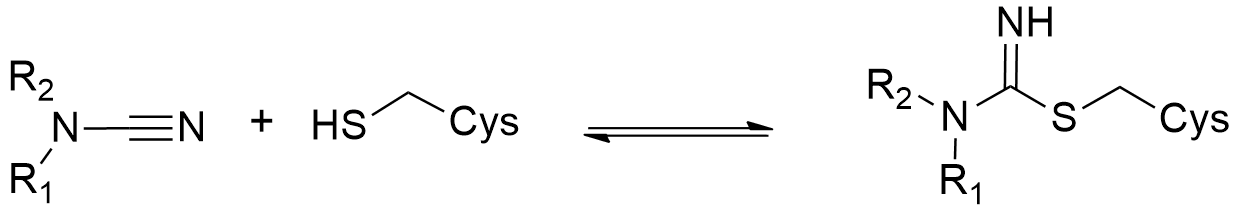
Cyanamide reversible binding to Cys-residues
We offer over 700 cyanamide compounds as dry powders and have designed a pre-plated library of 240 compounds. Covalent Screening N-nitriles Library is available in a pre-plated format at 10 mM concentration in DMSO. It can be purchased either as a standalone set or as a sublibrary of Covalent Screening Library.
Download SD files
761 compounds for cherry-picking
920K compounds
Plated libraries in stock:
Examples of Cyanamides
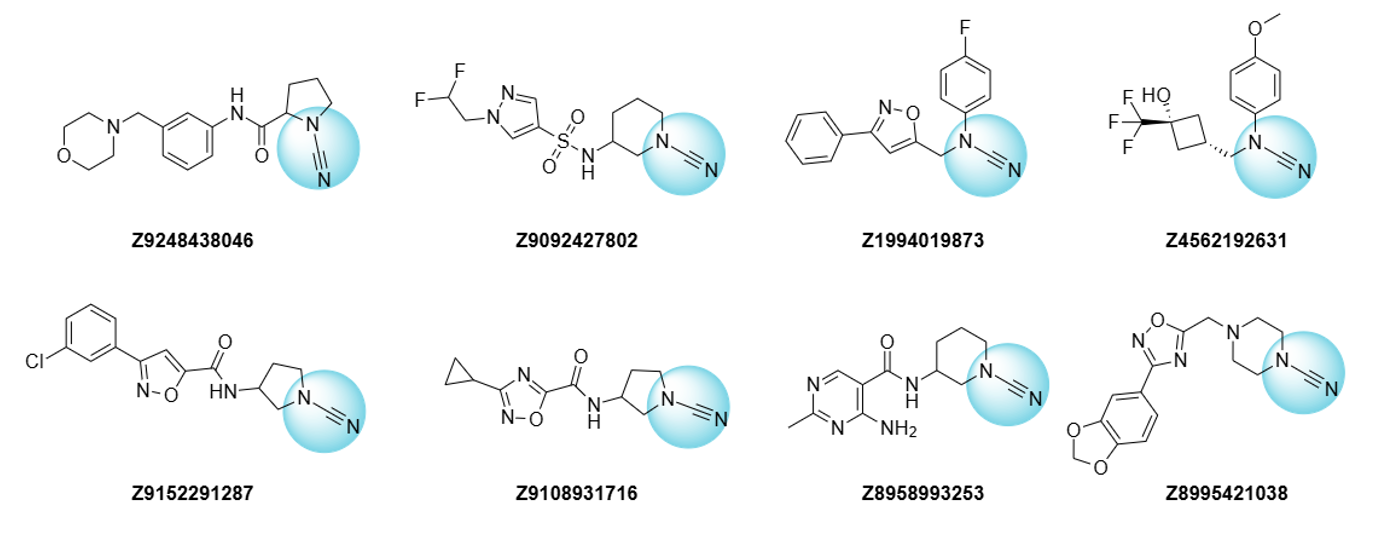
Synthetic approaches
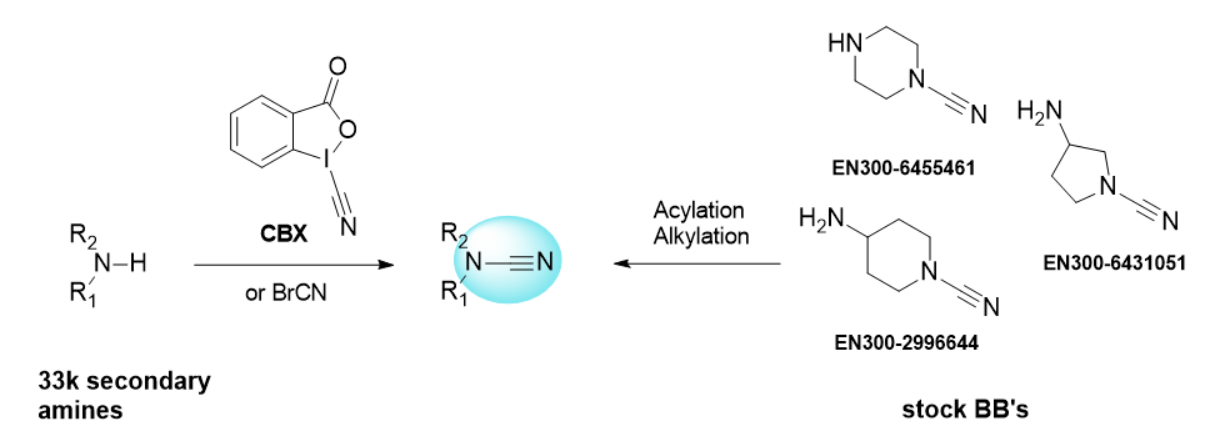
Two complementary synthetic strategies have enabled the development of a diverse range of cyanamide compounds. On the one hand, alkylation or acylation of building blocks with pre-installed cyanamide warhead has enabled the generation of many diverse compounds. On the other hand, direct cyanation of Enamine`s vast amine collection using a novel and convenient reagent (CBX) has provided access to structurally diverse cyanamides. Validation of various synthetic approaches, combined with access to our extensive building block stock, allowed us to create a virtual library of 920,000 cyanamide compounds. Compounds from this library can be synthesized within 3–4 weeks by our parallel chemistry department.
A Subset of Enamine REAL Compounds, Valuable Addition to The REAL/xREAL Space
Enamine unREAL® Space is a new, original extension to Enamine REAL compounds.
This collection is broad and diverse, opening a much larger access to synthetically accessible chemical space for drug discovery. All compounds from the unREAL Space are available in the shortest possible time thanks to well-established and validated synthesis protocols. The unREAL Compounds are assembled using 24 unique, well-validated synthetic protocols not included in the REAL/xREAL Space.
Key Facts
- Over 70.9B new, unique compounds that complement the REAL/xREAL Space
- Over 38B compounds compliant with Ro5, and over 33B compounds compliant with Veber's rule
- Shipping from 4 weeks
- Over 80% of compounds delivered
- All compounds undergo extensive quality control by 1H NMR and LC-MS, and purity is guaranteed to be over 90%.
Applications
- Hit finding
- SAR and hit-to-lead
- Compound library extension
Novelty
The unREAL Compounds are an innovative dataset of screening compounds that unlocks access to billions of previously unknown, unique structures. Enamine specialists continuously develop new and original building blocks, expanding access to an infinite chemical space for drug discovery. In most cases, Enamine provides the ability to supply these compounds without publishing them in their in-stock catalogs and online databases (Reaxys, SciFinder, Pubchem, etc.).
Diversity
The unREAL Space is built on 620 million Bemis-Murcko scaffolds that are unique and not present in the REAL/xREAL Space. This incredible diversity of scaffolds is made possible by utilizing over 86,700 unique building blocks assembled via 24 synthetic protocols.
Accessibility
The unREAL Space and its subsets are available upon request. Please send us an inquiry to