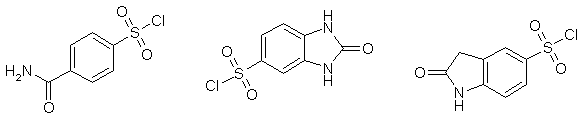
Quite often hydrogen bonding interaction of a ligand amide group with a protein has its functional consequences. To a great extend protein - ligand binding occurs via hydrogen bonding. In the search for new hits and leads it is very important in combinatorial synthesis to use the least possible number of synthetic steps. Reactions of sulfonic acid chlorides are well studied and quite often used in nucleophilic substitution reaction.
Development of building blocks that have both a sulfonic acid chloride group and nucleophilic amide group is not a trivial task as these two groups could potentially react with each other. Nevertheless there is a high interest of customers in sulfonyl chlorides bearing unsubstituted amide group. In our program directed to synthesis of new sulfonyl chlorides we carefully designed, synthesized and selected those sulfonyl chlorides that are stable for storage, but still sufficiently reactive for nucleophilic substitution. These building blocks could be used in the synthesis of targeted and focused libraries.
Few examples are given below: