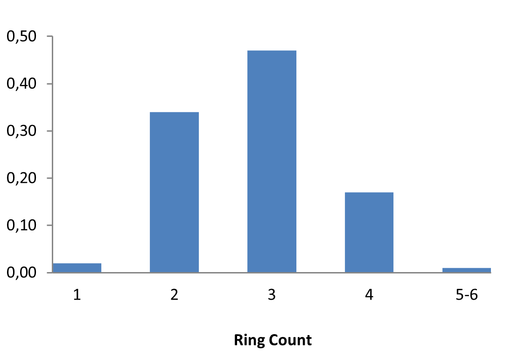
Expanding Covalent Warhead Space via SNAr Chemistry
960 compounds
Cysteine-targeting covalent inhibitors are currently dominated by acrylamide and chloroacetamide warheads. In contrast, interest among medicinal chemists in heteroaromatic electrophiles is steadily increasing. Recently, 2‑Sulfonylpyrimidines have gained particular attention due to their ability to undergo cysteine‑selective covalent modification via a nucleophilic aromatic substitution (SNAr) mechanism under mild, physiologically relevant conditions. These features make 2-sulfonylpyrimidines particularly attractive aromatic warheads for the development of next‑generation targeted covalent inhibitors. To support the MedChem community with easy access to these emerging covalent binders, we have developed a 960-compound Covalent 2-Sulfonylpyrimidine Library, available as 10 mM solutions in DMSO.
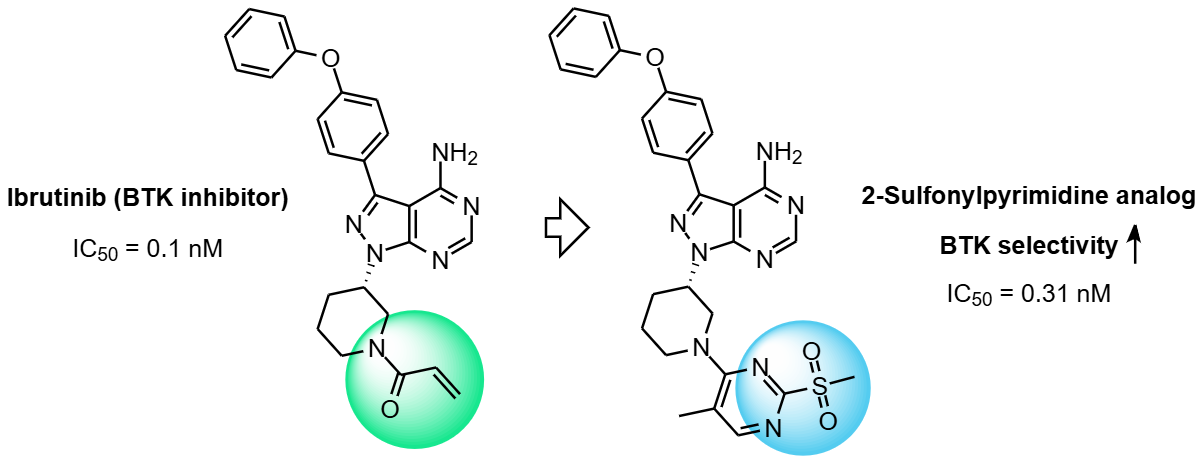
2‑Sulfonylpyrimidine-based Ibrutinib analogs have already been successfully applied in the development of potent and selective BTK covalent inhibitors. These compounds have been shown to function as effective acrylamide surrogates with comparable cellular potency and, importantly, improved kinome selectivity.
Typical Formats
Catalog No.
CSPL-960-0-Z-2
Compounds
960
1 plate
Amount
≤ 300 nL of 2 mM DMSO solutions
Plates and formats
1536 well, Echo Qualified 001-6969 (LP-0400), first four and last four columns empty, 960 compounds per plate
Price
Catalog No.
CSPL-960-10-Y-10
Compounds
960
3 plates
Amount
10 µL of 10 mM DMSO solutions
Plates and formats
384 well, Echo Qualified LDV microplates 001-12782 (LP-0200), first and last two columns empty, 320 compounds per plate
Price
Catalog No.
CSPL-960-50-Y-10
Compounds
960
3 plates
Amount
50 μL of 10 mM DMSO solutions
Plates and formats
384 well, Greiner Bio-One microplates 781280, first and last two columns empty, 320 compounds per plate
Price
*We will be happy to provide our library in any other most convenient for your project format. Please select among the following our standard microplates: Greiner Bio-One 781270, 784201, 781280, 651201 or Echo Qualified 001-12782 (LP-0200), 001-14555 (PP-0200), 001-6969 (LP-0400) or send your preferred labware. Compounds pooling can be provided upon request.
Key features
- Cysteine-selective SNAr reactivity
- Irreversible covalent binding
- Tuned reactivity
- Hydrolytically stable
Library design
Design of the library focused on functionalization of the 2-sulfonylpyrimidine core using structurally diverse building blocks. Robust synthetic transformations such as arylation and amide bond formation were used to access the final compounds. These synthetic routes provide efficient access to analogs via parallel chemistry. To modulate SNAr electrophilic reactivity, we introduced various electron-donating and electron-withdrawing substituents on the pyrimidine core.
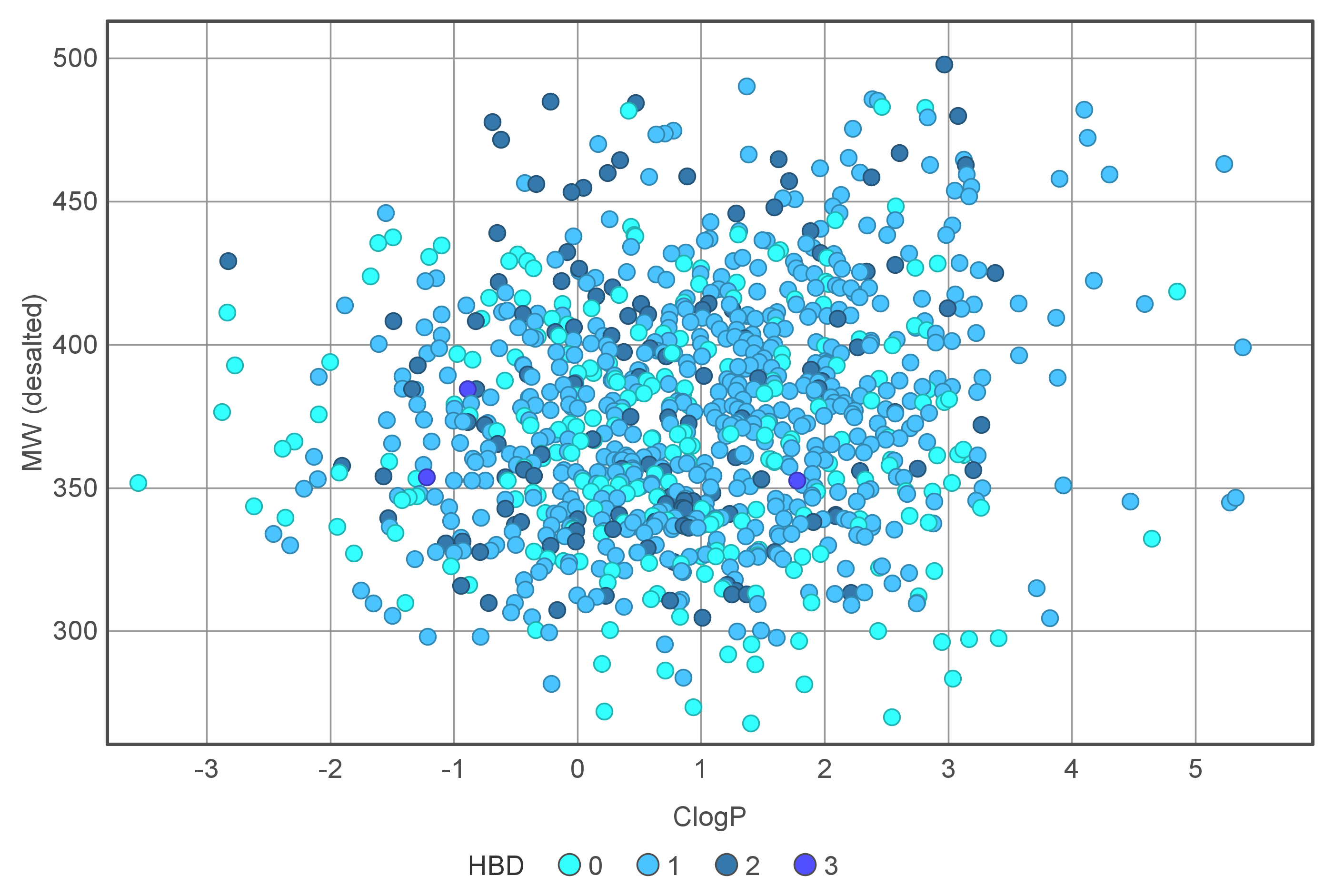
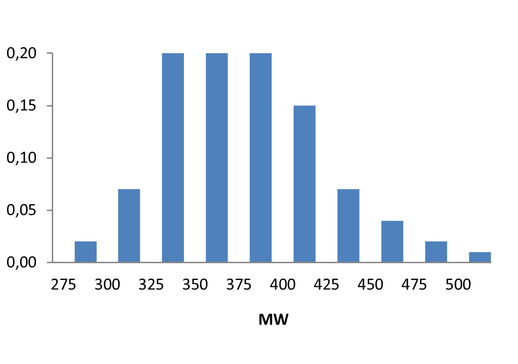
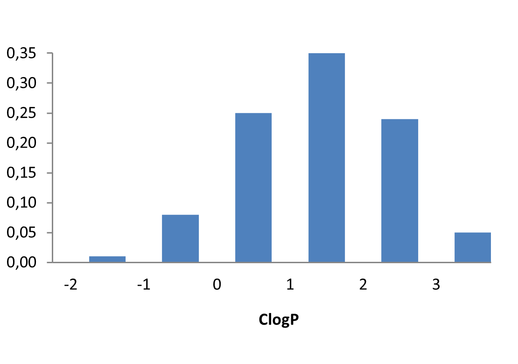
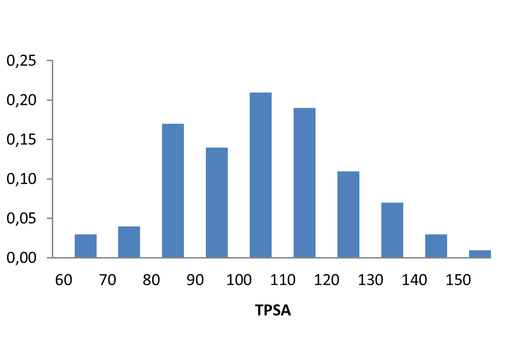
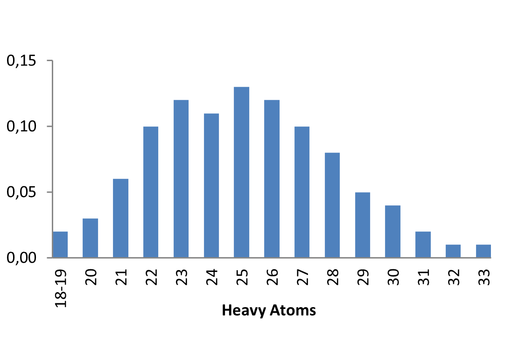
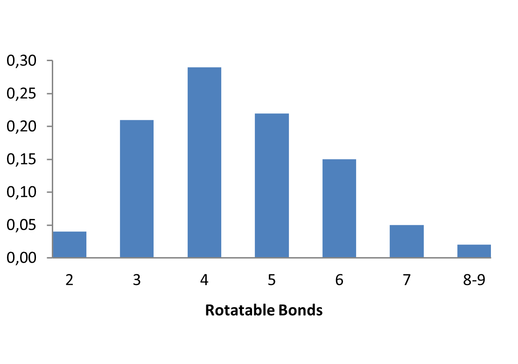
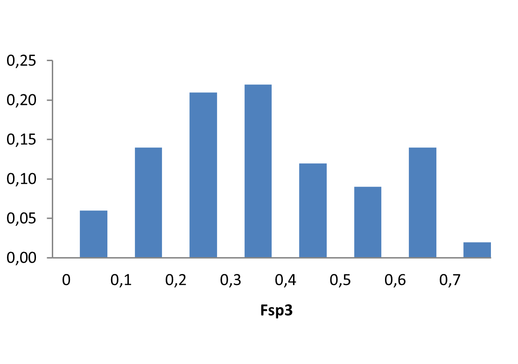
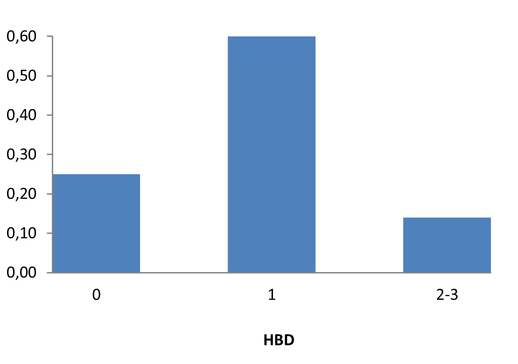
Medicinal chemistry filters were applied during library design to remove undesirable motifs and optimize drug-like properties. Final compound selection for the pre-plated library ensured balanced coverage across key physicochemical parameters (MW, LogP, HBD etc), supporting broad applicability in screening campaigns.
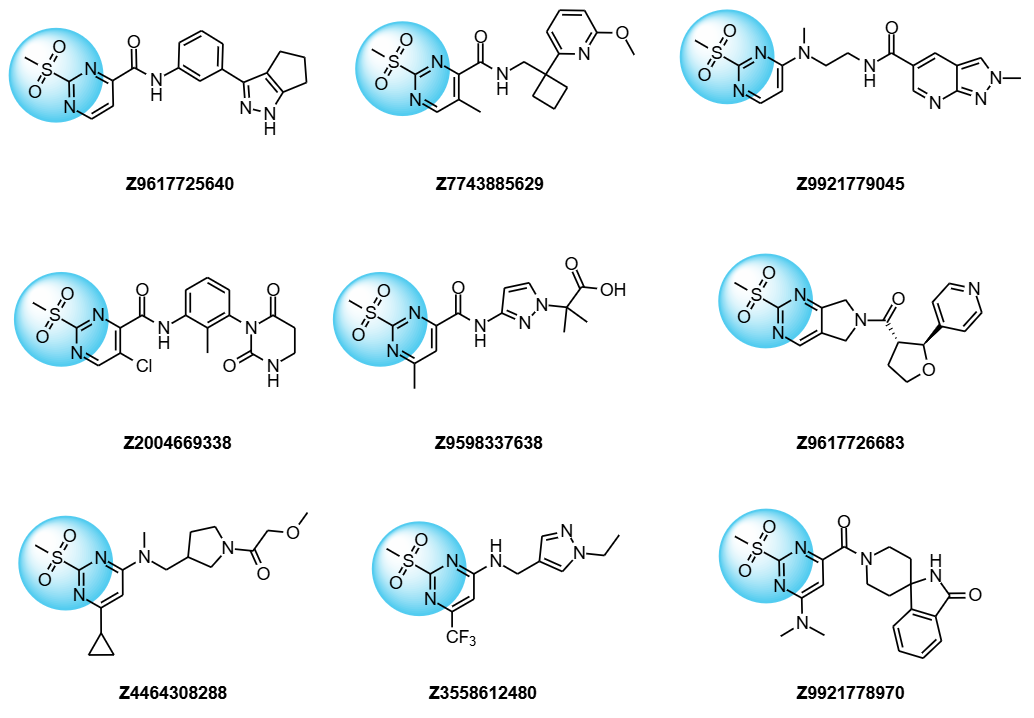
Examples of compounds from Covalent 2-Sulfonylpyrimidine Library
Covalent 2-Sulfonylpyrimidine Library cLogP/MW/HBD distribution