CAS 3033096-03-3, Cat. No EN300-53610231
Methylation reagent
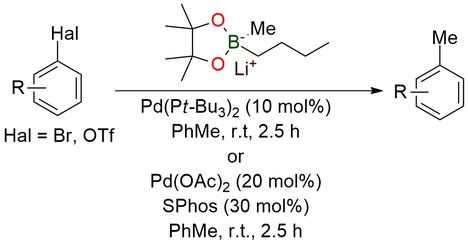
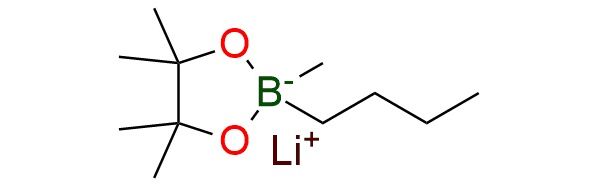
Lithium 2-butyl-2,4,4,5,5-pentamethyl-1,3,2-dioxaborolan-2-uide (“SuzukiMe”) is a pre-activated lithium dialkyl boronate reagent designed for base-free Pd-catalyzed methylation of aryl electrophiles1. Unlike conventional boronic acids or esters, it exists as a tetra-coordinate boron “ate” complex, which significantly enhances nucleophilicity and enables direct transmetalation without external hydroxide or alkoxide base. This allows efficient cross-coupling of aryl bromides and triflates under mild conditions (room temperature, short reaction times), overcoming the limitations of traditional methylation reagents, which often require harsh conditions or exhibit low reactivity. The reagent demonstrates broad substrate scope, including functionalized arenes, heterocycles, and complex drug-like molecules, with excellent tolerance toward base-sensitive groups (esters, amides, heterocycles) and high chemoselectivity (e.g., bromide vs triflate). It is bench-stable, a free-flowing solid that can be stored for extended periods without loss of activity.
Synonyms: lithium 2-butyl-2,4,4,5,5-pentamethyl-1,3,2-dioxaborolan-2-uide; lithium(1+) 2-butyl-2,4,4,5,5-pentamethyl-1,3,2-dioxaborolan-2-uide
Selected publication
-
SuzukiMe: A Tactically Mild.
Harbit, R. C.; Leão, L. P. M. de O.; Korobko, S. V.; Sotnik, S. O.; Volochnyuk, D. M.; Ryabukhin, S. V.; Smith, J. M. ChemRxiv 2026. DOI: 10.26434/chemrxiv.15001572/v1