CAS 3060319-85-6, Cat. No EN300-52863357
Reagent for decarboxylative trifluoroethylation
![N'-[2,2,2-trifluoroethylidene]-4-(trifluoromethyl)benzene-1-sulfonohydrazide](/images/Reagents/EN300-52863357_Scheme.jpg)
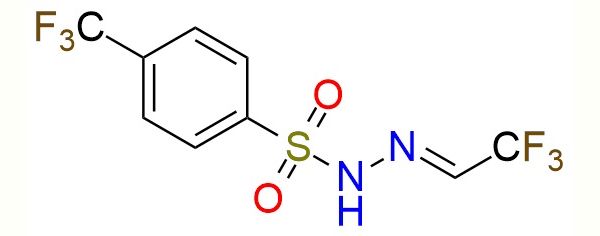
N'-[2,2,2-trifluoroethylidene]-4-(trifluoromethyl)benzene-1-sulfonohydrazide (para-TFHZ-Tfs) is a bench-stable sulfonyl hydrazone derived from trifluoroacetaldehyde, developed as a formal trifluoroethyl (–CH₂CF₃) transfer reagent under visible-light conditions1. Under photocatalytic conditions, alkyl radicals—most commonly generated by decarboxylation of aliphatic carboxylic acids - add to the C=N bond of the sulfonyl hydrazone. The resulting alkylated hydrazide then undergoes light-induced diazene fragmentation, extruding N₂ and a sulfinate to furnish trifluoroethylated products. This two-step, one-pot sequence cleanly converts R-CO₂H into R-CH₂CF₃, providing a distinct retrosynthetic disconnection compared to classical CF₃ methods. The reagent is not a CF₃ source but a C1-homologative CF₃ equivalent, formally delivering –CH₂CF₃. The methodology demonstrates broad applicability to primary, secondary, and tertiary aliphatic carboxylic acids, as well as cyclic amines, heterocycles, amino acids, peptides, and complex drug-like molecules. The protocol tolerates halogens, esters, amides, carbamates, alcohols, protected amines, and heteroaromatics and is suitable for late-stage functionalization. The reagent shows high chemoselectivity for radical addition and favors the desired radical fragmentation pathway under light, suppressing competing anionic fluoride-elimination processes. The reagent can also undergo [3+2] annulation with 4-nitroisoxazoles to form trifluoromethylpyrazolo [3,4-d]isoxazoles2.
Synonyms: para-TFHZ-Tfs; (E)-N'-(2,2,2-trifluoroethylidene)-4-(trifluoromethyl)benzenesulfonohydrazide; benzenesulfonic acid, 4-(trifluoromethyl)-, 2-(2,2,2-trifluoroethylidene)hydrazide; 4-(trifluoromethyl)benzenesulfonic acid 2-(2,2,2-trifluoroethylidene)hydrazide
Selected publications
-
A C1-Homologative Trifluoromethylation: Light-Driven Decarboxylative Trifluoroethylation of Carboxylic Acids.
Belnome, F.; Pulcinella, A.; Bonciolini, S.; Lepori, M.; Datsenko, O. P.; He, Z.; Gasparetto, M.; Mykhailiuk, P. K.; de Bruin, B.; Noël, T. J. Am. Chem. Soc 2026. DOI: 10.1021/jacs.5c21423
-
The [3+2] Annulation of in Situ Formed Trifluorodiazoethane with 4-Nitroisoxazoles: Access to Trifluoromethylpyrazolo[3,4-d]Isoxazoles.
Wang, H.; Meng, L. Q.; Niu, W.; Yang, W. Q.; Ou, Y.; Lin, Y. W. Chemical Communications 2024, 60 (85), 12425–12428. DOI: 10.1039/d4cc03826f