CAS 2831416-43-2, Cat. No EN300-46967415
C1 radical synthon
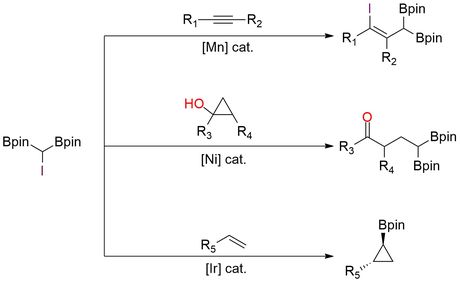
(Diborylmethyl)iodide, or CHI(Bpin)₂, is a bench-stable α-iodo gem-diboryl reagent that functions as a versatile C1 radical synthon1. The presence of two Bpin groups strongly stabilizes the adjacent carbon-centered radical, enabling controlled reactivity under mild catalytic conditions. In photocatalytic chemistry, CHI(Bpin)₂ is used as a radical cyclopropanation reagent for alkenes2. Under visible-light photoredox catalysis (typically fac-Ir(ppy)₃ with CsF), it undergoes single-electron reduction to generate an α-diboryl radical, which adds to alkenes followed by polar cyclization to afford 1,2-substituted cyclopropyl boronic esters in good yields. The reaction shows high chemo- and diastereoselectivity and tolerates a wide range of functional groups, including halides, esters, amides, alcohols, heterocycles, and complex natural-product derivatives. Beyond alkenes, CHI(Bpin)₂ also participates in atom-transfer radical addition (ATRA) to alkynes under manganese catalysis and visible light, delivering γ-iodo-allylic gem-diboronates with high Z/E selectivity (up to ~96:4). These products are valuable synthetic linchpins, as the C–I bond and Bpin groups can be selectively transformed via cross-coupling, deborylation, oxidation, or homologation. In transition-metal catalysis, CHI(Bpin)₂ serves as an ambiphilic α-bimetalloid radical precursor in nickel-catalyzed C(sp³)–C(sp³) coupling reactions, such as ring-opening of cyclopropanols to give γ-carbonyl gem-diboronates3. This chemistry highlights the reagent’s ability to engage in radical–metal crossover pathways and further underscores the synthetic versatility of gem-diboryl motifs.
Synonyms: 2,2'-(iodomethylene)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane); 1,3,2-dioxaborolane, 2,2′-(iodomethylene)bis[4,4,5,5-tetramethyl- ; CHI(Bpin)₂; 2-[iodo(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane; (diborylmethyl)iodide
Selected publications
-
Synthesis of γ-Iodo-Allylic Diboronic Esters via Atom Transfer Radical Addition of (Diborylmethyl)Iodide to Alkynes.
Zhang, K.; Zhang, J.; He, Q.; Hu, J.; Jing, S. Org. Lett. 2025. DOI: 10.1021/acs.orglett.5c00730
-
Photocatalyzed Borylcyclopropanation of Alkenes with a (Diborylmethyl)Iodide Reagent.
Hu, J.; Tang, M.; Wang, J.; Wu, Z.; Friedrich, A.; Marder, T. B. Angewandte Chemie - International Edition 2023, 62 (38). DOI: 10.1002/anie.202305175
-
Nickel-Catalyzed C(Sp3)–C(Sp3) Coupling of Cyclopropanols via α-Bimetalloid Radicals: Access to γ-Carbonyl Gem-Bis(Boronates) and Gem-Silylboronate Esters.
Zhu, L.; Zhang, X.; Wang, H. Org. Lett. 2025, 27 (40), 11408–11413. DOI: 10.1021/acs.orglett.5c03886