CAS 931109-52-3, Cat. No EN300-21834963
Reagent for amination and aziridination
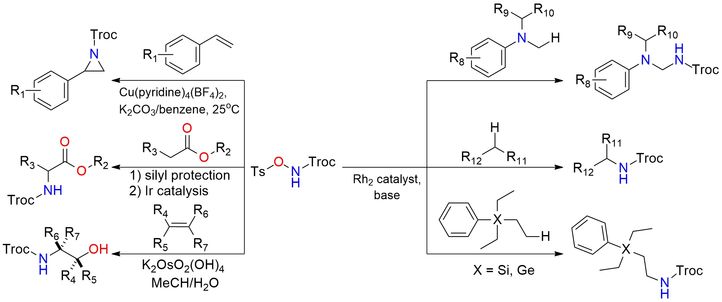
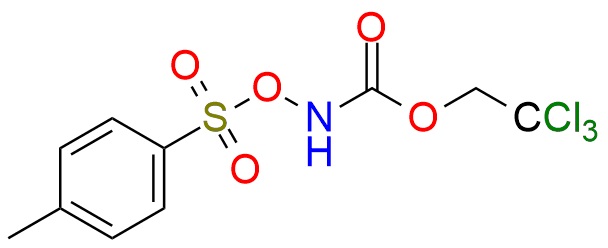
2,2,2-Trichloroethyl N-tosyloxycarbamate (Troc-NH-O-Ts) is a bench-stable N-sulfonyloxycarbamate reagent used as a metal–nitrene precursor for C–H amination and aziridination reactions1. Under basic conditions in the presence of dirhodium catalysts, it generates reactive metal–nitrene species that enable direct C(sp³)–H amidation via insertion, forming Troc-protected amines with moderate to good yields and broad functional-group tolerance2,3. The reagent is specifically designed to suppress intramolecular side reactions (no δ-C–H bonds), enabling efficient intermolecular C–H functionalization of benzylic, aliphatic, and heteroatom-adjacent positions. It is also effective in aziridination of alkenes and related nitrene-transfer processes, typically under mild conditions (base, metal catalyst), providing protected nitrogen-containing products4. Additionally, N-(tosyloxy)carbamates can serve as preformed nitrogen sources in oxyamination of alkenes, affording vicinal amino alcohol derivatives with good regioselectivity5. Ir-catalyzed amidation of silyl ketene acetals by the reagent proceeds to the formation of α-amido esters6. α-Amidation applies to both α-aryl and α-alkyl esters. A key advantage is the Troc protecting group, which can be readily removed under mild reductive conditions to give free amines, making the reagent highly practical for synthesis and late-stage functionalization.
Synonyms: [(2,2,2-trichloroethoxy)carbonyl]azanyl 4-methylbenzenesulfonate; 2,2,2-trichloroethyl (tosyloxy)carbamate; 2,2,2-trichloroethyl N-tosyloxycarbamate; Troc-NH-O-Ts; 2,2,2-trichloroethyl N-[(4-methylbenzenesulfonyl)oxy]carbamate; benzenesulfonic acid, 4-methyl-, [(2,2,2-trichloroethoxy)carbonyl]azanyl ester
Selected publications
-
2,2,2‐Trichloroethyl‐ N ‐Tosyloxycarbamate.
Lebel, H.; Charbonnier, C. Encyclopedia of Reagents for Organic Synthesis 2025, 1–4. DOI: 10.1002/047084289X.rn01011.pub2
-
Dirhodium-Catalyzed Chemo-and Site-Selective C-H Amidation of N, N-Dialkylanilines.
Chen, G.; Arai, K.; Morisaki, K.; Kawabata, T.; Ueda, Y. Synlett 2021, 32 (7), 728–732. DOI: 10.1055/a-1334-6450
-
ß-Silicon-Effect-Promoted Intermolecular Site-Selective C(Sp3)-H Amination with Dirhodium Nitrenes.
Ninomiya, R.; Arai, K.; Chen, G.; Morisaki, K.; Kawabata, T.; Ueda, Y. Chemical Communications 2020, 56 (43), 5759–5762. DOI: 10.1039/d0cc00959h
-
N-Tosyloxycarbamates as Reagents in Rhodium-Catalyzed C-H Amination Reactions.
Huard, K.; Lebel, H. Chemistry - A European Journal 2008, 14 (20), 6222–6230. DOI: 10.1002/chem.200702027
-
Osmium-Catalyzed Vicinal Oxyamination of Alkenes by N-(4-Toluenesulfonyloxy)Carbamates.
Masruri; Willis, A. C.; McLeod, M. D. Journal of Organic Chemistry 2012, 77 (19), 8480–8491. DOI: 10.1021/jo301372y
-
Iridium-Catalyzed Amidation of In Situ Prepared Silyl Ketene Acetals to Access α-Amino Esters.
Gwon, Y.; Lee, M.; Kim, D.; Chang, S. Org. Lett 2022, 24 (4), 1088–1093. DOI: 10.1021/acs.orglett.1c04376