J. Fluorine Chem. , 2010, 131 (2), 221-223
DOI: 10.1016/j.jfluchem.2009.07.017
Mykhailiuk P. K.; Radchenko D. S.; Komarov I. V.
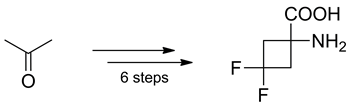
A new fluorinated analogue of 1-aminocyclobutane-1-carboxylate (ACBC) – 1-amino-3,3-difluorocyclobutanecarboxylic acid (6) – has been synthesized in six steps from acetone. The key step of the synthesis is a transformation of a ketone group into the CF2-group using morpholino-sulphur trifluoride.
Mykhailiuk P. K.; Radchenko D. S.; Komarov I. V.
J. Fluorine Chem. 2010, 131 (2), 221-223
DOI: 10.1016/j.jfluchem.2009.07.017
