J. Org. Chem. , 2012, 77 (2), 1174-1180
DOI: 10.1021/jo2022235
Tymtsunik A. V.; Bilenko V. A.; Kokhan S. O.; Grygorenko O. O.; Volochnyuk D. M.; Komarov I. V.
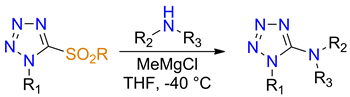
An approach to the synthesis of 1-alkyl-5-((di)alkylamino)tetrazoles by nucleophilic substitution in 1-alkyl-5-sulfonyltetrazoles with anions generated from the primary or secondary amines was developed. Tolerance of the method to the presence of some functional groups (i.e., protected amine) in both components of the reaction was demonstrated. Obtained tetrazoles are promising building blocks for the design of peptide surrogates, in particular, for replacement approaches of alkyl urea derivatives.
Tymtsunik A. V.; Bilenko V. A.; Kokhan S. O.; Grygorenko O. O.; Volochnyuk D. M.; Komarov I. V.
J. Org. Chem. 2012, 77 (2), 1174-1180
DOI: 10.1021/jo2022235
