As I already wrote in my previous post , we at Enamine are constantly working on extending our REAL concept (where REAL is for REadily AccessibLe compounds). Basically, this concept allows for the generation of a virtual chemical space based on the experimentally validated synthetic accessibility. Until now the REAL Space has comprised 15.5 billion make-on-demand molecules and is currently the largest offer of commercially available compounds. It was shown that this chemical space is especially useful in combination with high-throughput virtual screening techniques (see Nature 2019, 566, 224–229 and Nature 2020, 580, 663–668 ); there is also a number of other ways to access it.
In a new paper just published in European Journal of Organic Chemistry , we for the first time describe application of the REAL principles to the generation of artificial macrocycle libraries. A very simple and robust chemistry based on the "build‐couple‐pair" diversity‐oriented (DOS) strategy was used to construct a virtual library of 180,000 macrocycles derived from 12,283 different scaffolds. The approach showed a 61% success rate under the parallel synthesis conditions, which is a very good result for the 4–5‐step reaction sequence including two chromatographic purifications. More than 40% compounds from the Macro Space bear an amino function suitable for the post‐pairing modification, thus giving a rise to at least a billion‐sized chemical space, as well as macrocyclic building blocks available on a gram scale.
The Macro Space will not be included into the current version of REAL Space but is available as a separate offer
See the full paper for more details.
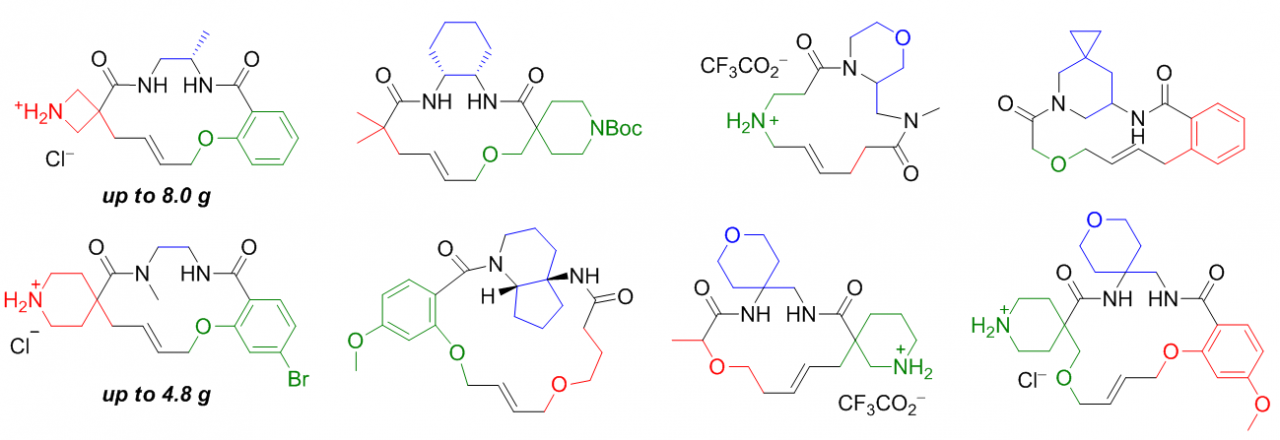
Examples of macrocycles
