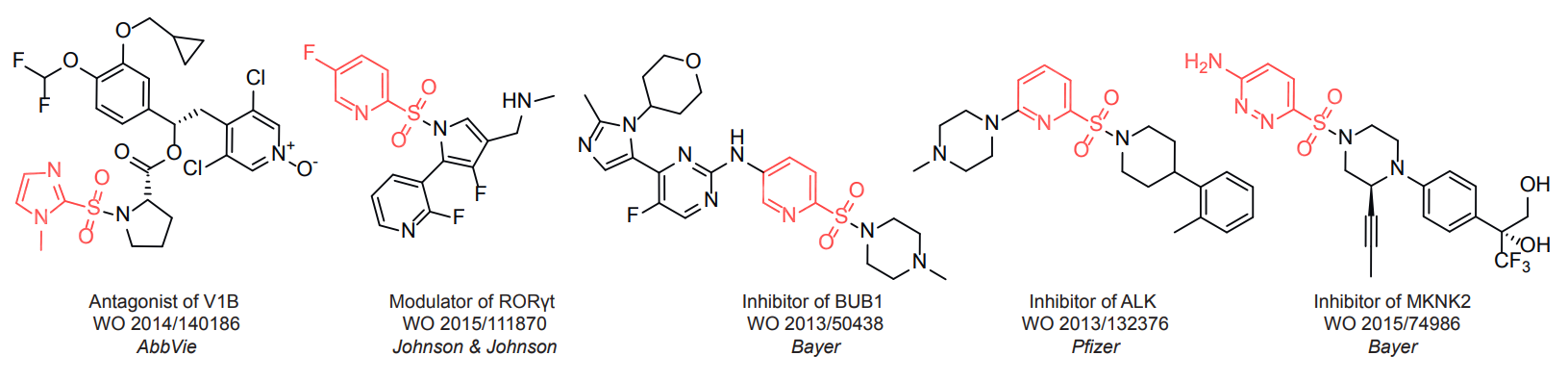
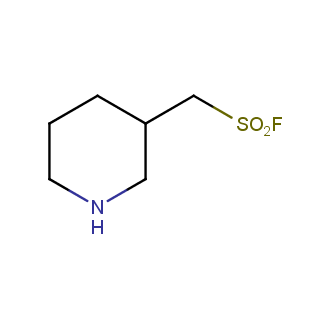
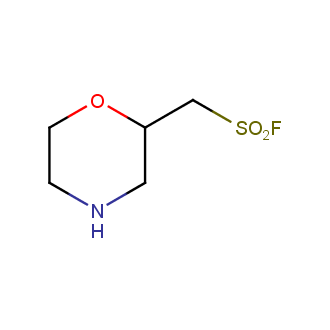
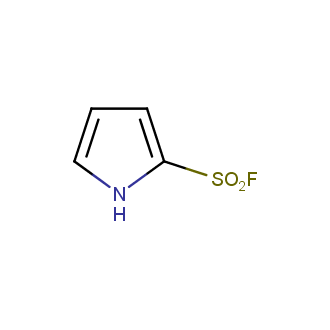
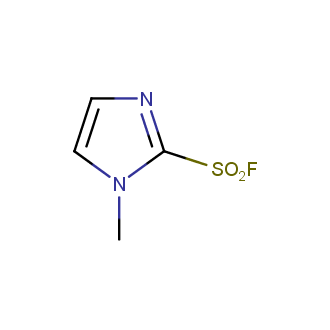
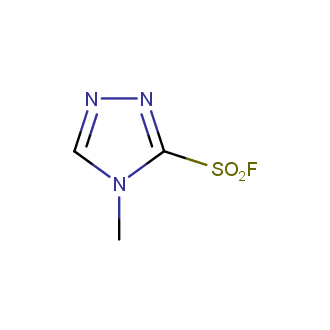
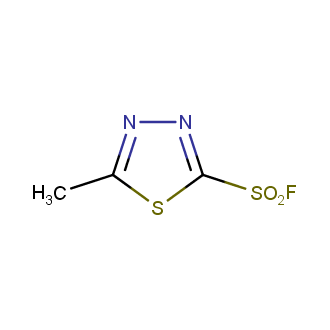
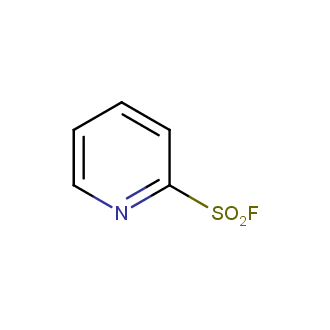
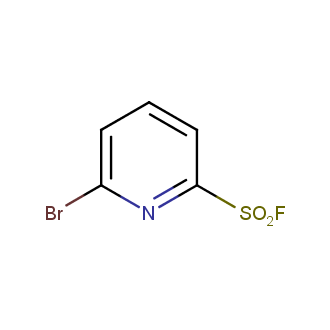
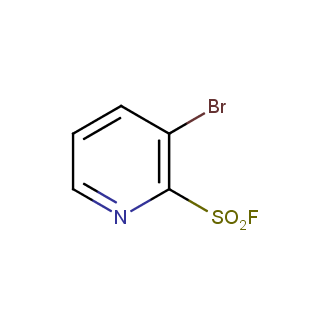
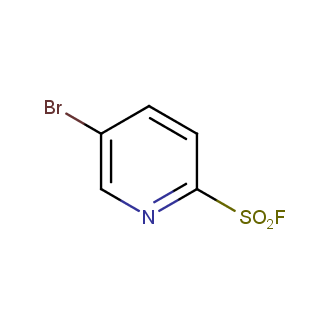
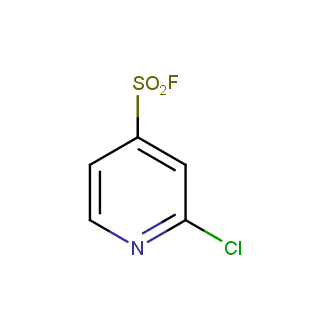
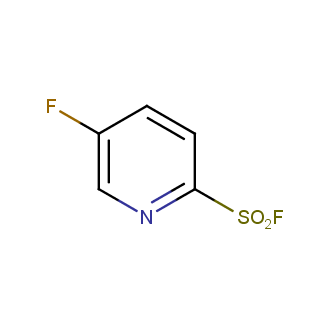
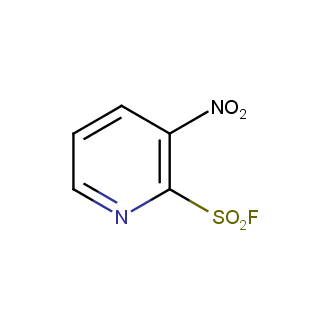
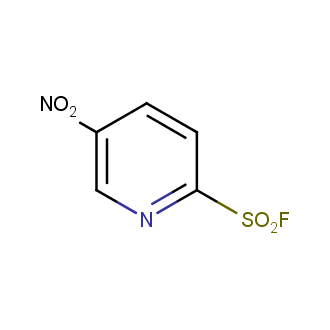
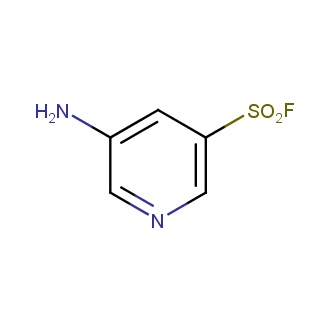
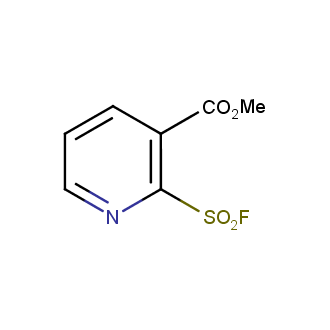
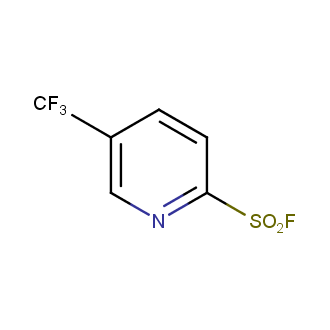
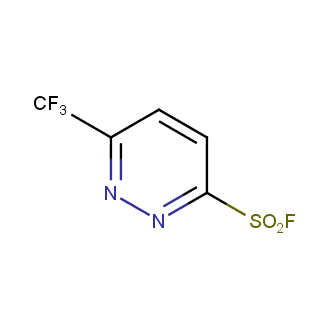
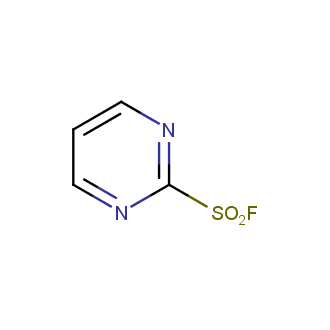
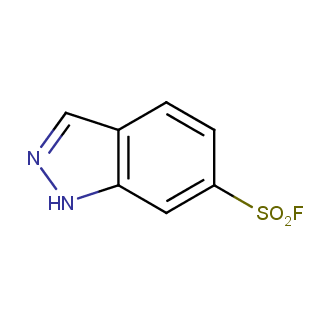
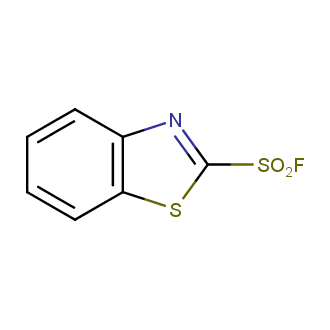
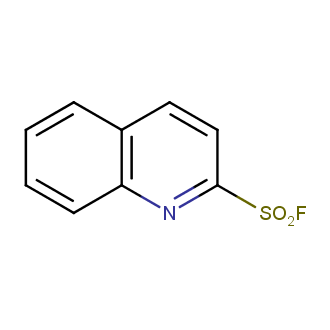
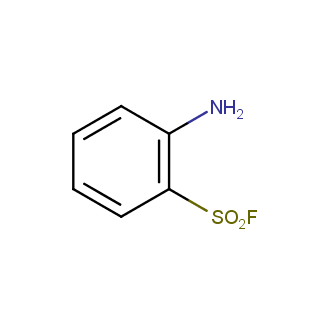
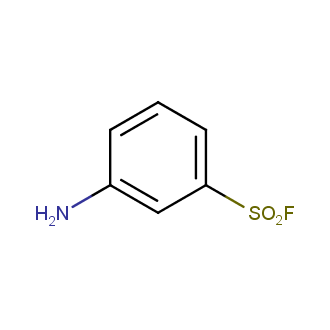
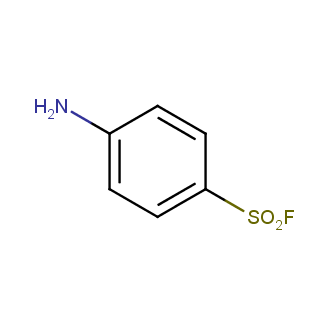
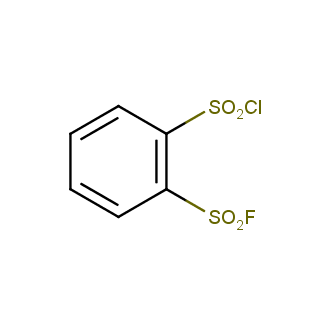
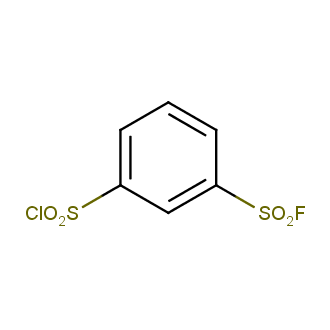
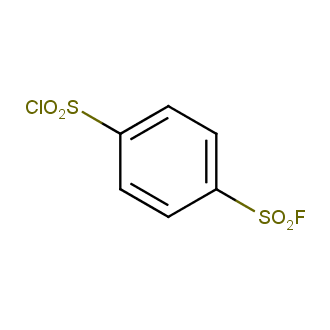
Sulfonyl chlorides (-SO2Cl) are widely used in medicinal chemistry and agrochemistry as precursors to pharmacologically important sulfonamides. Many sulfonyl chlorides with heteroaromatic substituents, however, are unstable due to SO2 extrusion. More stable sulfonyl fluorides (-SO2F) in many cases are the only option to synthesize the desired sulfonamides. They are less reactive, so that they might even have a free aliphatic amino groups in their structure. Besides unique monofunctional sulfonyl fluorides, Enamine offers a wide array of scaffolds and linker compounds.
The fluoride rescues the lead:

- Stable to moisture
- Improved selectivity
- Orthogonal reactivity to many other functional groups
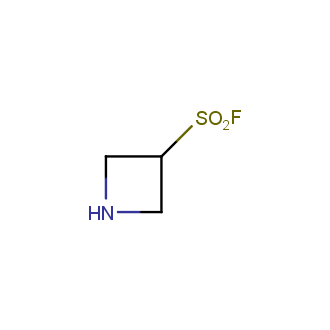
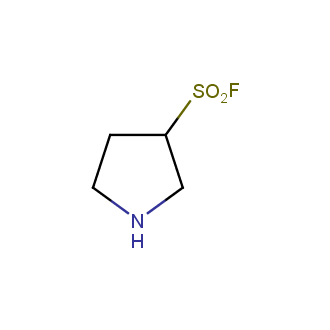
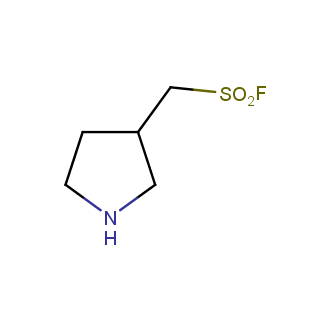
- Scaffolds
-SO2F probes in chemical biology
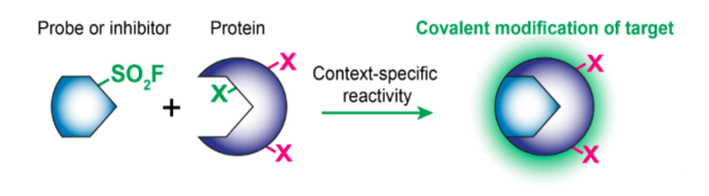
The SO2F group covalently binds to the residues of serine, threonine, tyrosine, lysine, cysteine, and histidine in proteins. Sulfonyl fluorides are widely used as chemical probes and covalent protein inhibitors.
Our offer:
>200 Sulfonyl fluorides (-SO2F) in gram amounts in stock. Custom synthesis of further analogues and compound libraries.