PLoS ONE , 2014, 9 (6), e99653
DOI: 10.1371/journal.pone.0099653
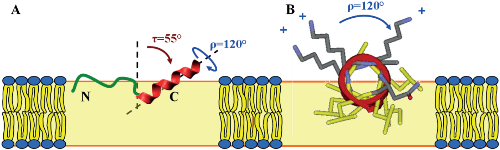
Structure analysis of the cell-penetrating peptide transportan 10 (TP10) revealed an exemplary range of different conformations in the membrane-bound state. The bipartite peptide (derived N-terminally from galanin and C-terminally from mastoparan) was found to exhibit prominent characteristics of (i) amphiphilic α-helices, (ii) intrinsically disordered peptides, as well as (iii) β-pleated amyloid fibrils, and these conformational states become interconverted as a function of concentration. We used a complementary approach of solid-state 19F-NMR and circular dichroism in oriented membrane samples to characterize the structural and dynamical behaviour of TP10 in its monomeric and aggregated forms. Nine different positions in the peptide were selectively substituted with either the L- or D-enantiomer of 3-(trifluoromethyl)-bicyclopent-[1.1.1]-1-ylglycine (CF3-Bpg) as a reporter group for 19F-NMR. Using the L-epimeric analogs, a comprehensive three-dimensional structure analysis was carried out in lipid bilayers at low peptide concentration, where TP10 is monomeric. While the N-terminal region is flexible and intrinsically unstructured within the plane of the lipid bilayer, the C-terminal α-helix is embedded in the membrane with an oblique tilt angle of ∼55° and in accordance with its amphiphilic profile. Incorporation of the sterically obstructive D-CF3-Bpg reporter group into the helical region leads to a local unfolding of the membrane-bound peptide. At high concentration, these helix-destabilizing C-terminal substitutions promote aggregation into immobile β-sheets, which resemble amyloid fibrils. On the other hand, the obstructive D-CF3-Bpg substitutions can be accommodated in the flexible N-terminus of TP10 where they do not promote aggregation at high concentration. The cross-talk between the two regions of TP10 thus exerts a delicate balance on its conformational switch, as the presence of the α-helix counteracts the tendency of the unfolded N-terminus to self-assemble into β-pleated fibrils.
Fanghänel S.; Wadhwani P.; Strandberg E.; Verdurmen W. P. R.; Bürck J.; Ehni S.; Mykhailiuk P. K.; Afonin S.; Gerthsen D.; Komarov I. V.; Brock R.; Ulrich A. S.
PLoS ONE 2014, 9 (6), e99653
DOI: 10.1371/journal.pone.0099653
