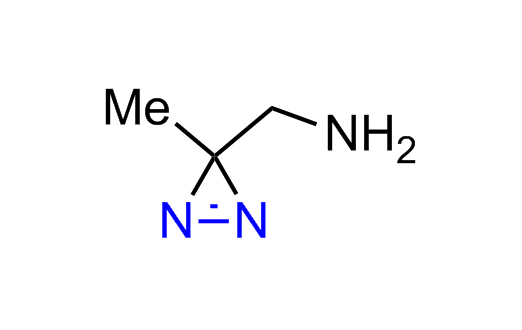
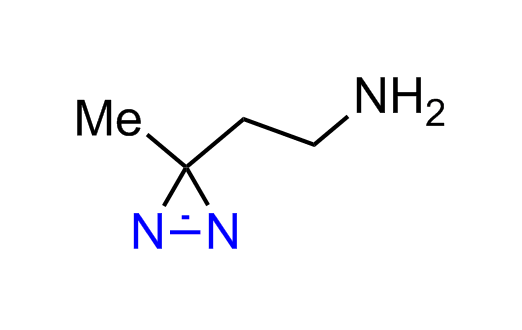
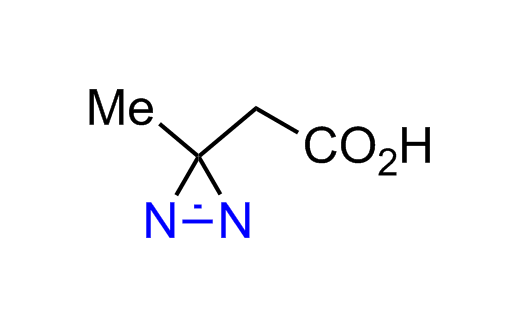
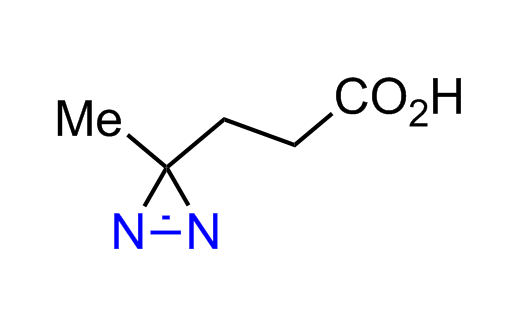
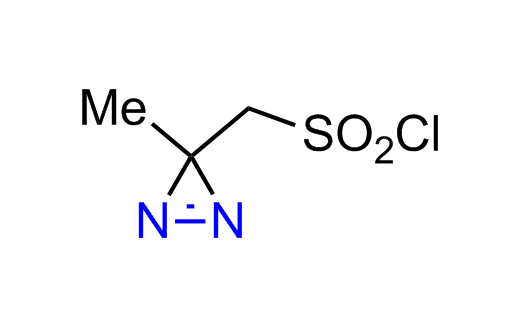
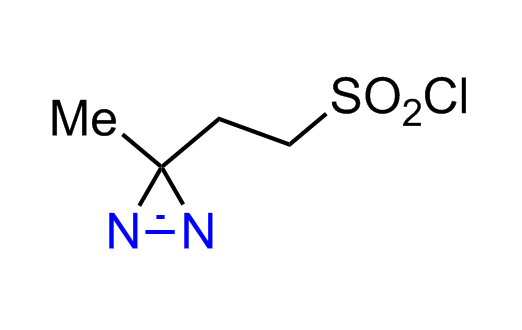
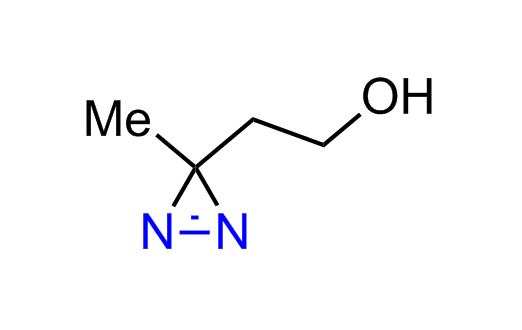
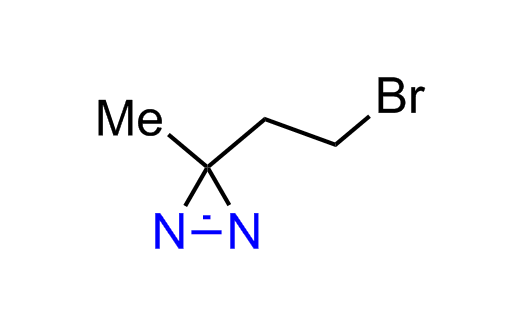
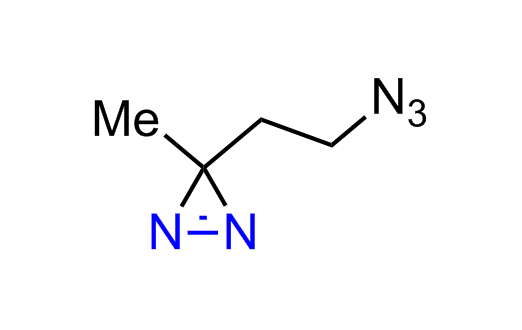
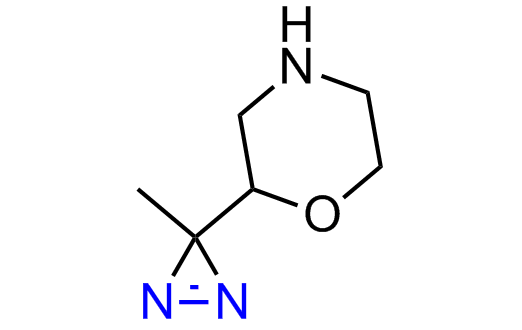
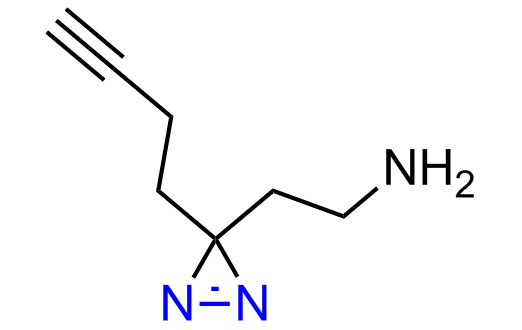
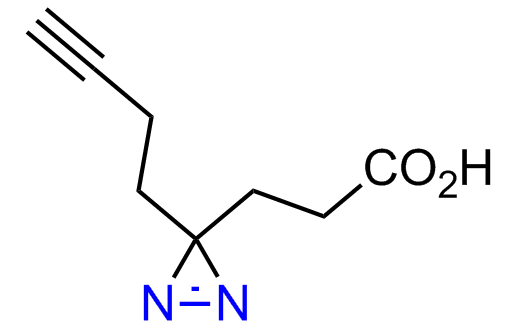
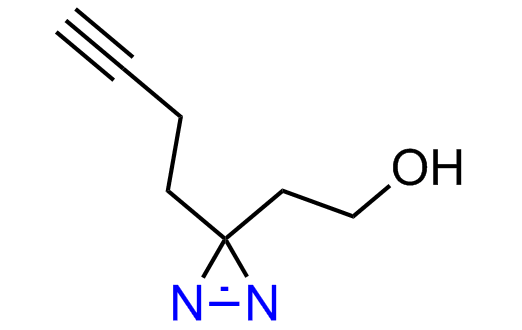
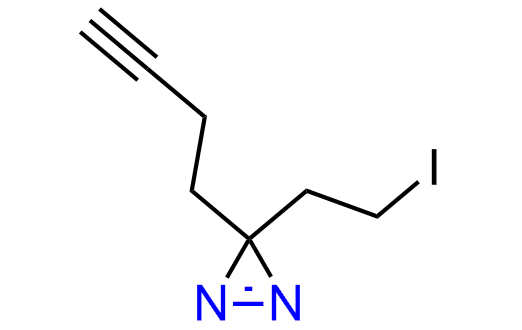
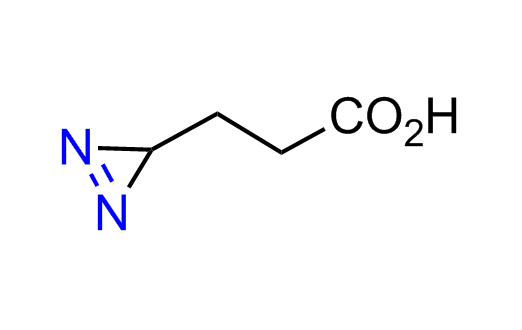
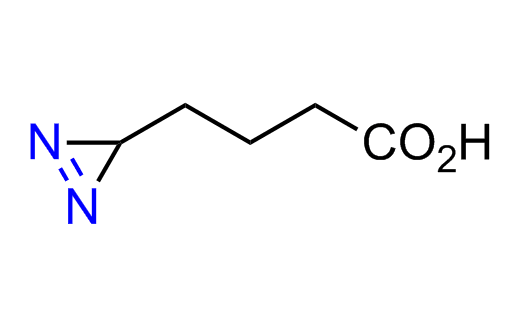
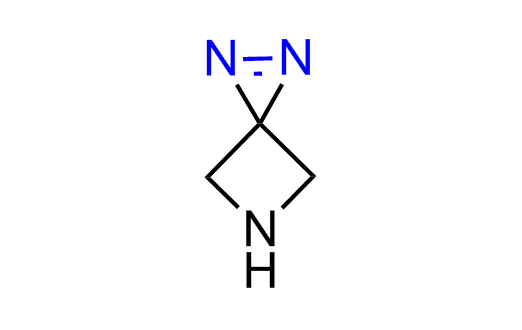
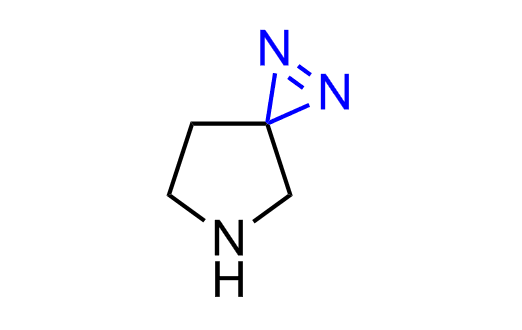
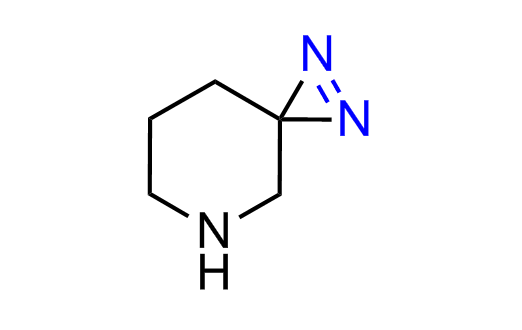
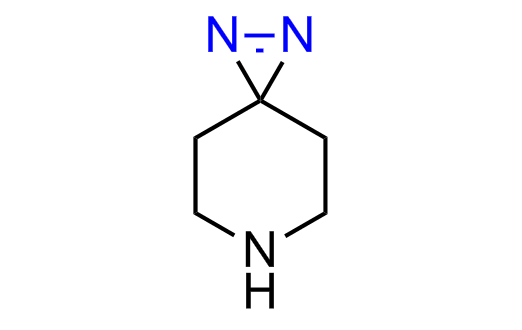
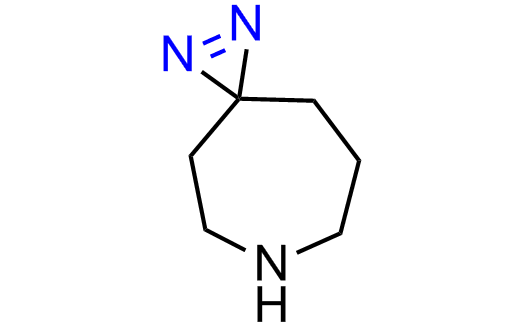
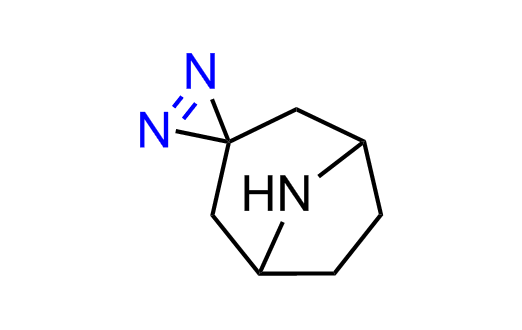
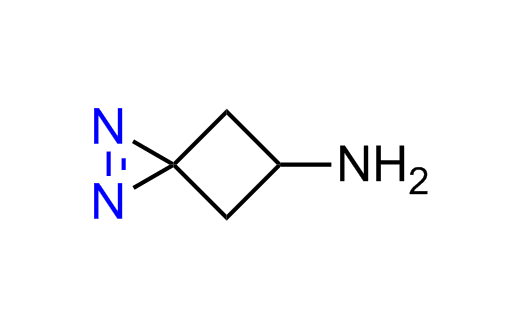
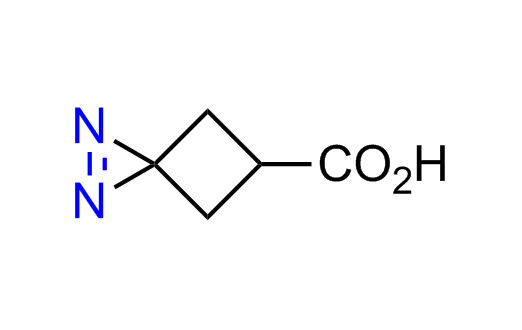
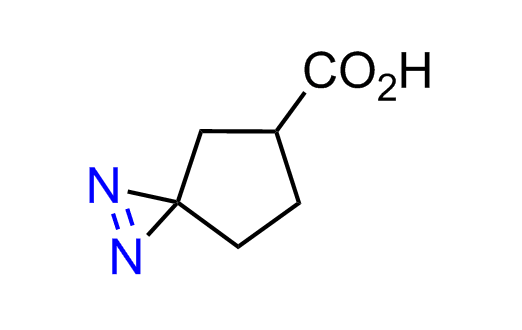
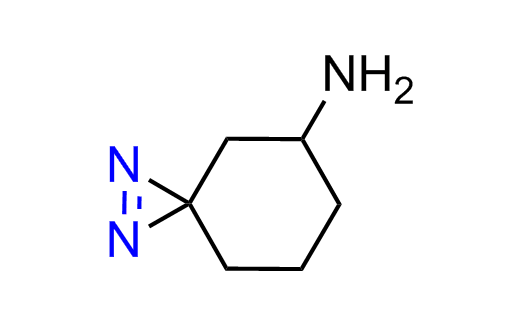
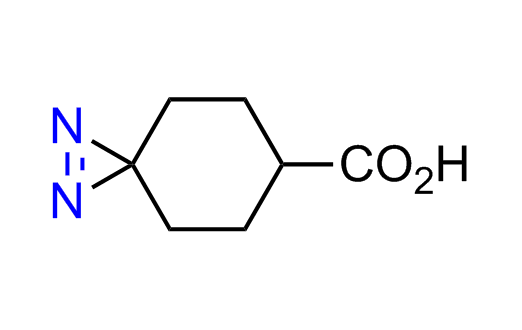
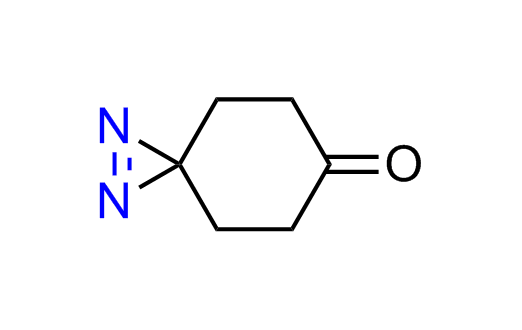
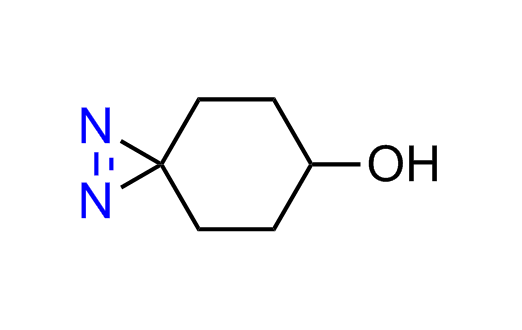
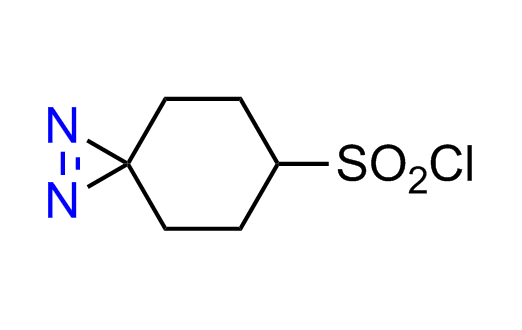
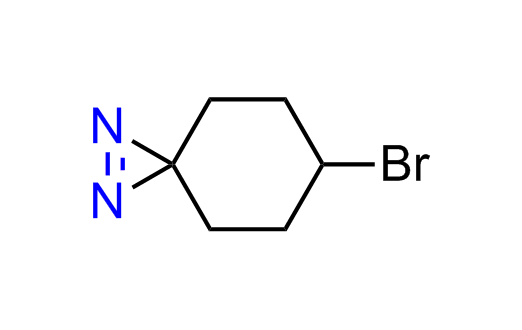
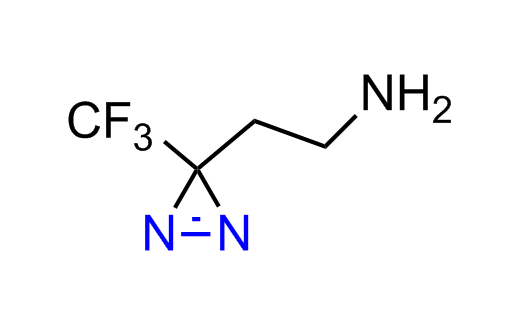
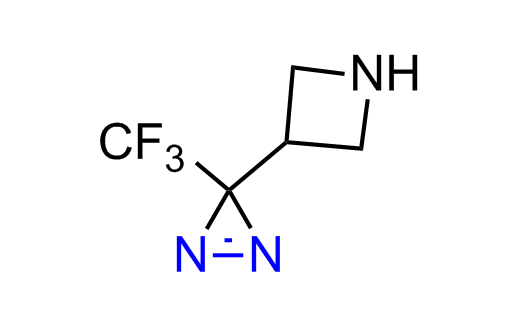
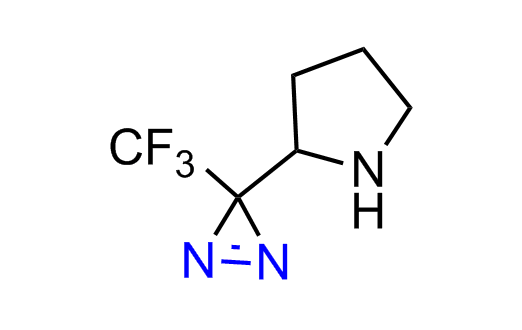
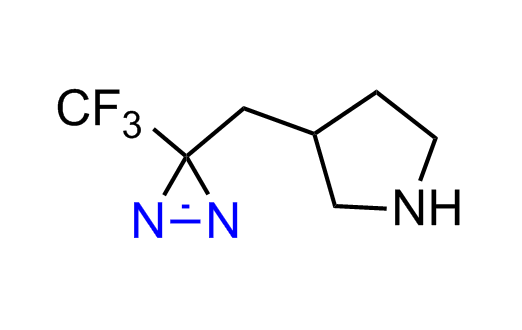
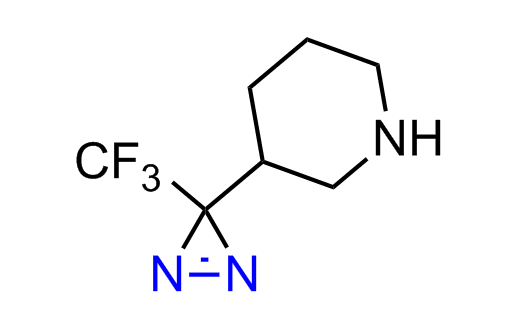
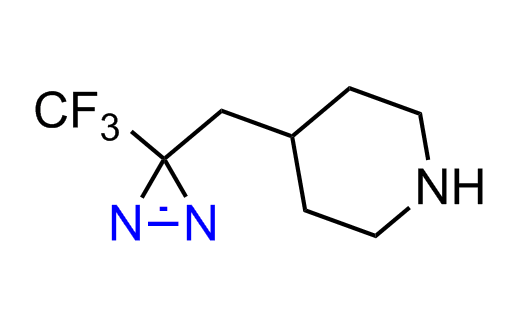
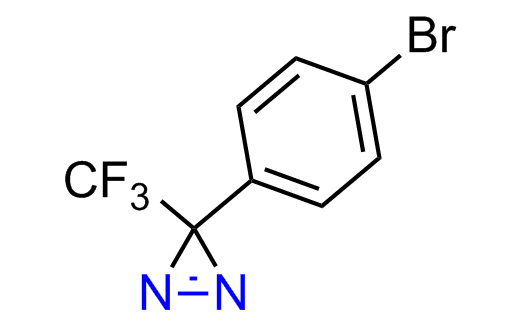
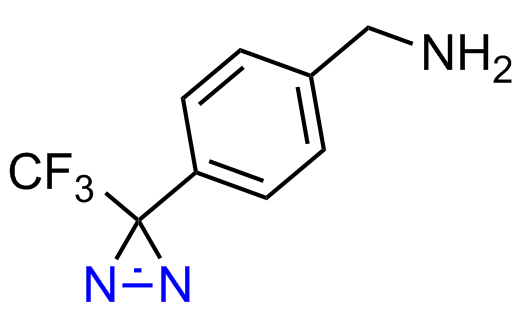
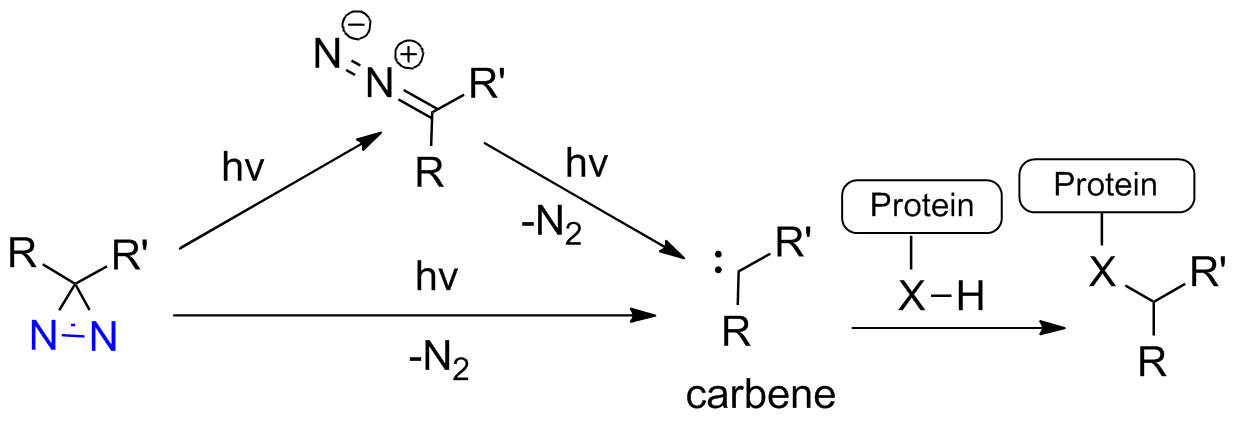
Diazirine is a smallest heterocycle that is stable in the dark, but forms reactive carbene upon irradiation with light. The formed carbenes smoothly insert into C-H, N-H, and O-H bonds. Therefore, diazirines are often used as photoreactive crosslinking reagents due to their small size, short period of irradiation, and stability toward nucleophiles. In this context, Enamine offers a library of diazirines for drug design.
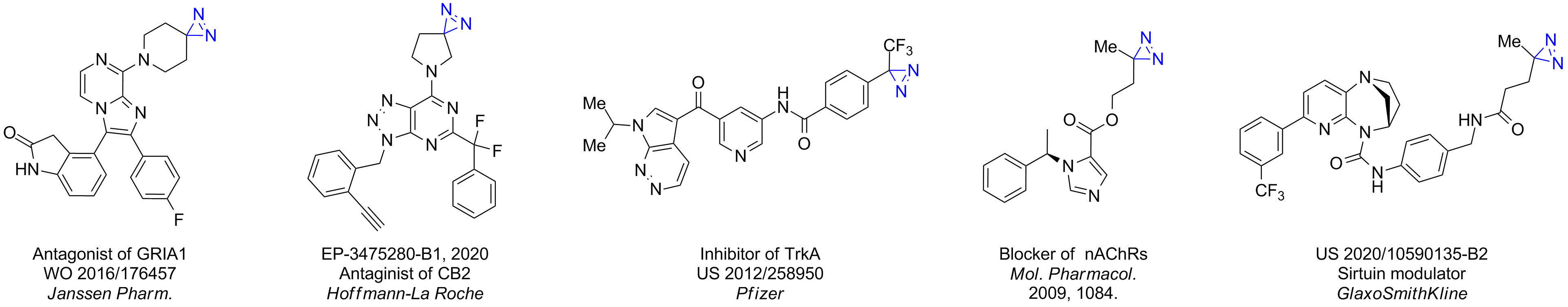
Case studies
Properties
- smallest photoreactive group;
- excitation at 355 nm;
- high chemical stability.
Upon irradiation of a ligand-target complex, a diazirine-containing ligand generates a reactive carbene that covalently binds the ligand to the target.
Download SD file
Download PDF file
We offer
More than 100 of diazirines from stock on a 5-10 g scale.