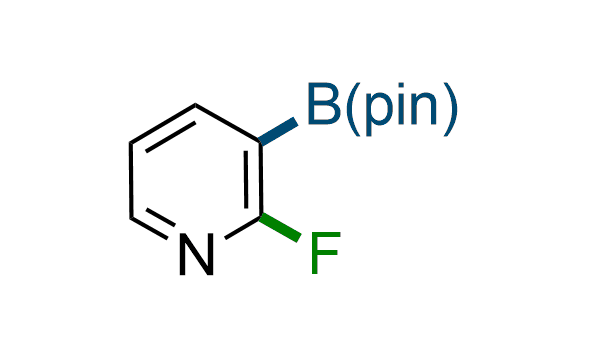
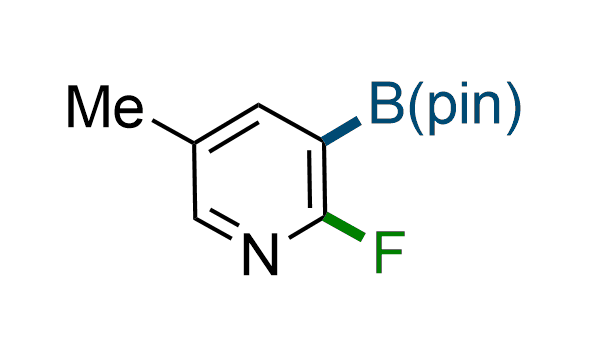
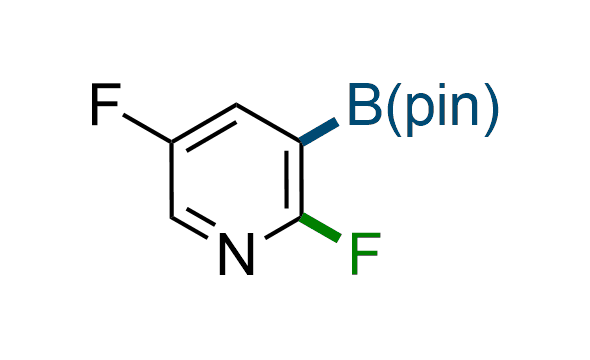
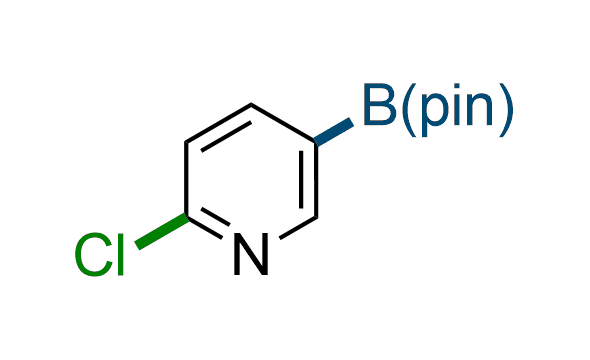
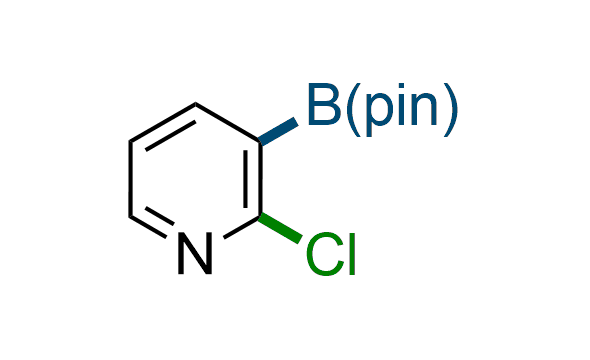
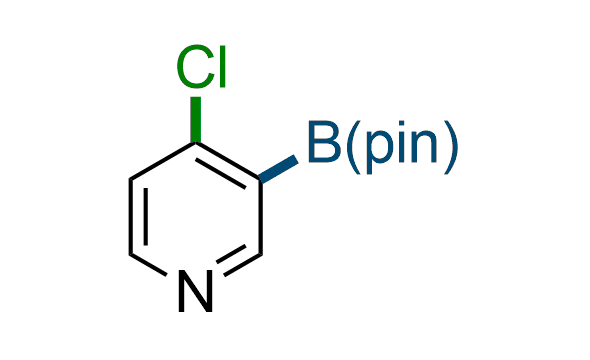
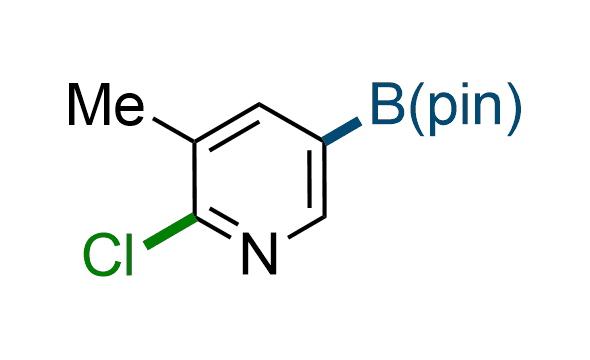
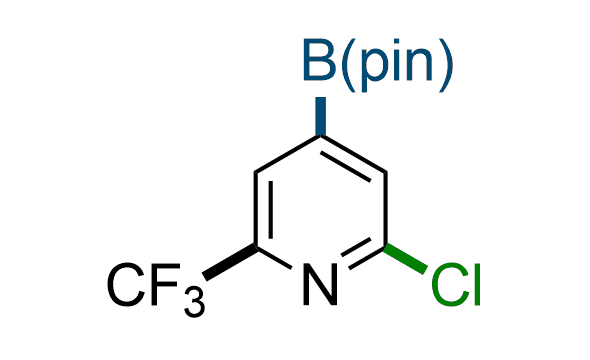
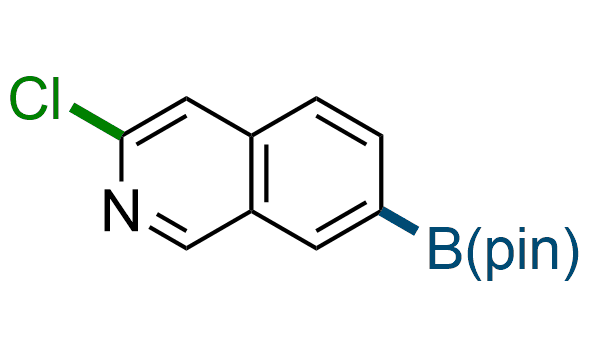
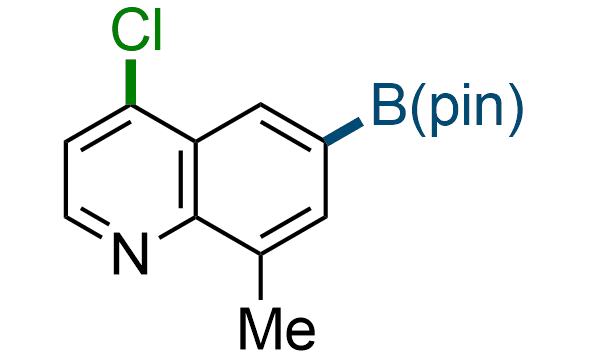
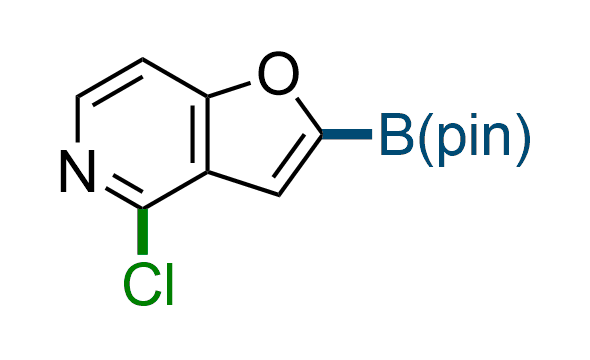
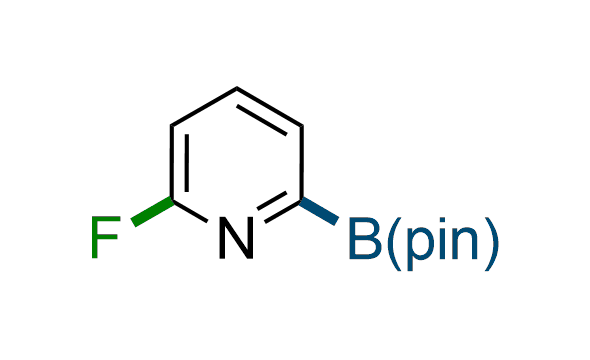The Suzuki–Miyaura cross-coupling of boronic acid derivatives is one of the most used reactions in organic and medicinal chemist's toolbox. The rapid advancement of this method resulted in its efficient application for the late-stage modification of biologically active substrates and construction of combinatorial libraries. Herein, we offer a library of heterocyclic boronic esters with active halogen atoms. They can be used for metal-mediated cross-coupling reactions followed by the stepwise a nucleophilic aromatic substitution (SNAr) to produce functionalized products.
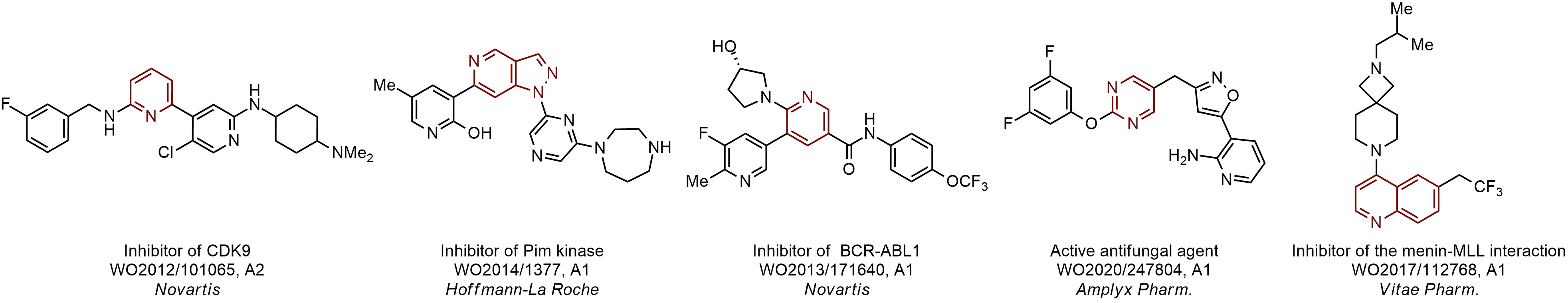
Case studies
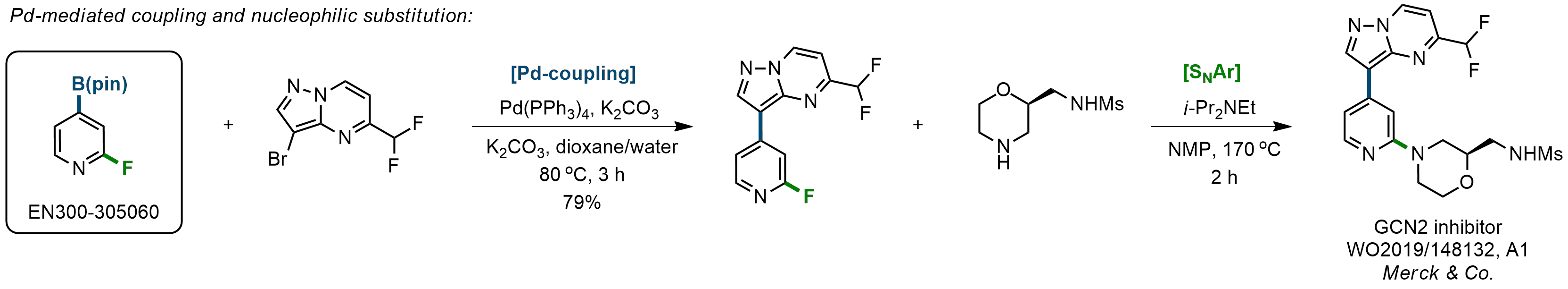
Download SD file
Download PDF file
We offer
>100 unique heterocyclic boronic esters on a 5-50 g scale from stock.