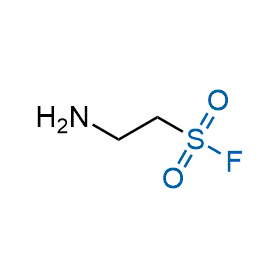
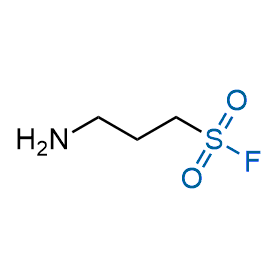
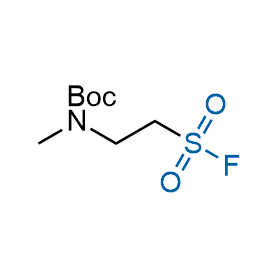
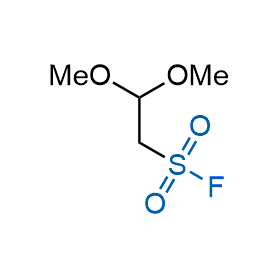
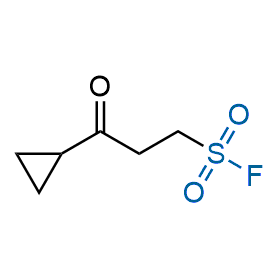
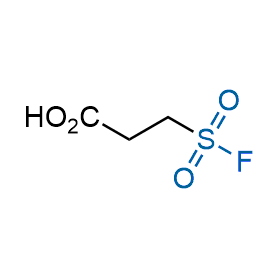
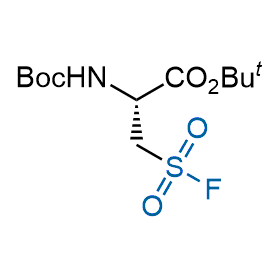
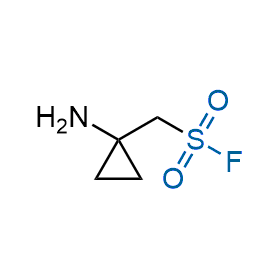
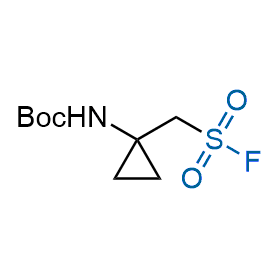
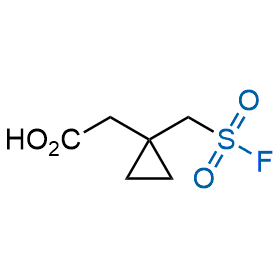
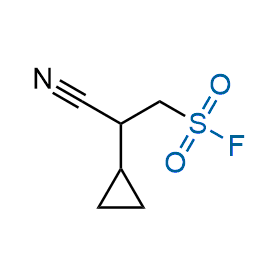
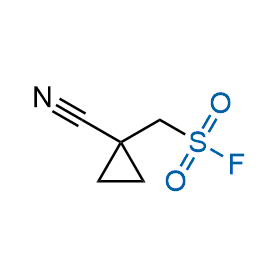
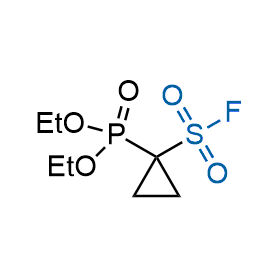
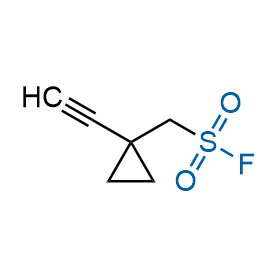
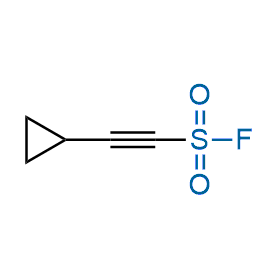
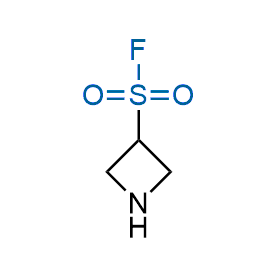
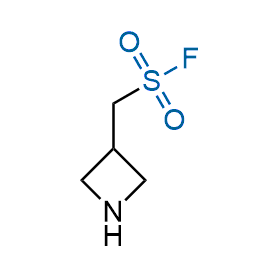
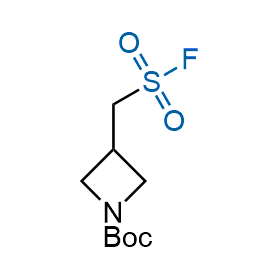
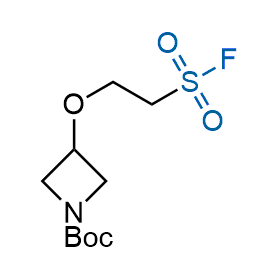
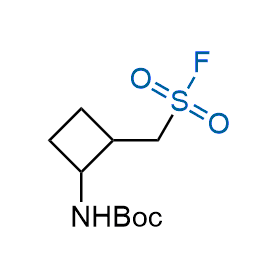
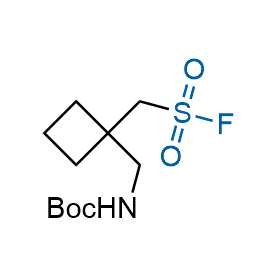
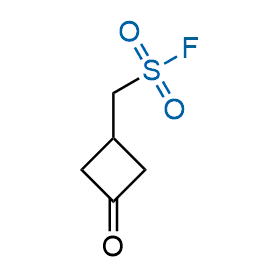
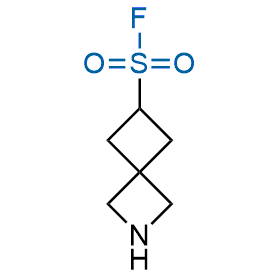
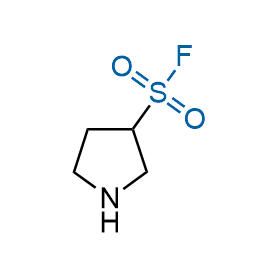
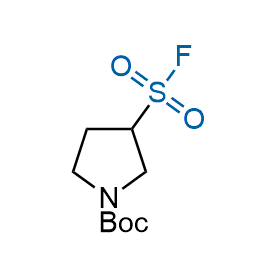
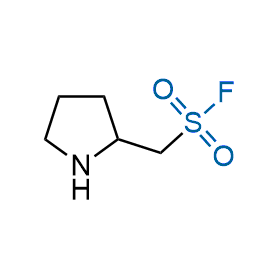
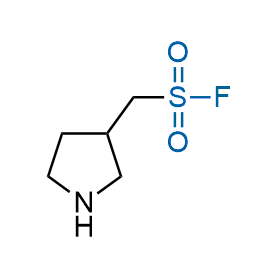
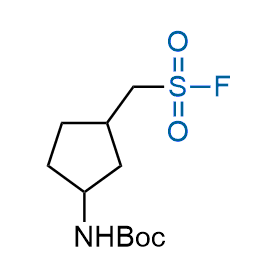
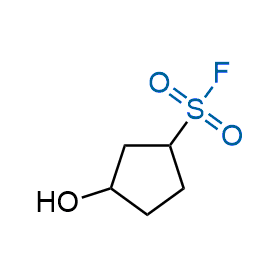
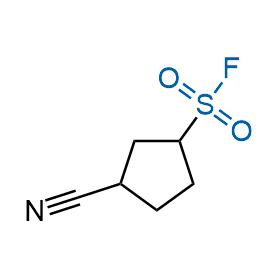
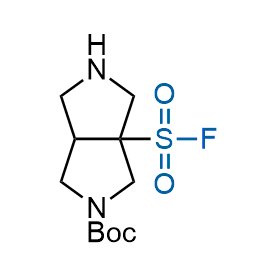
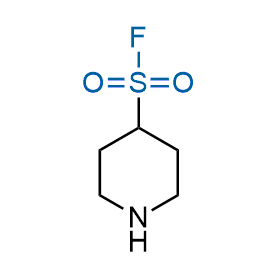
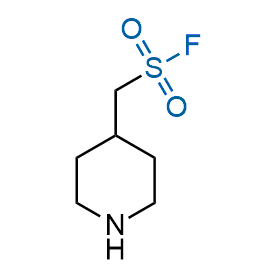
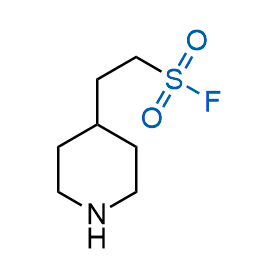
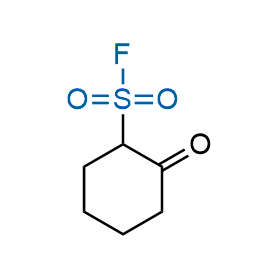
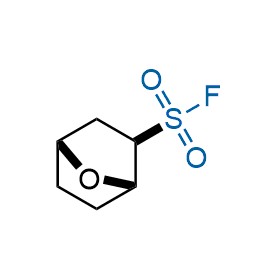
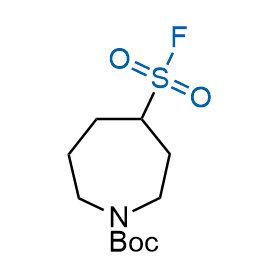
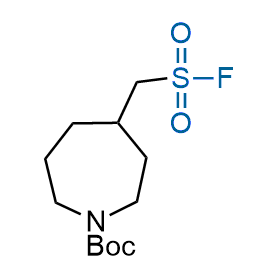
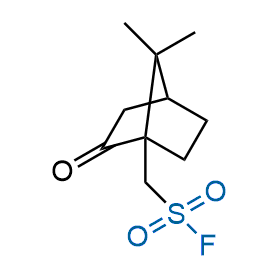
Following the original success of the alkyne-azide click chemistry, the sulfur (VI) fluoride exchange (SuFEx) has gained a wide recognition as a new family of click transformations. Aliphatic sulfonyl fluorides are excellent SuFEx agents owning to their moderate reactivity. These substances tolerate many other functional groups as well as handling in aqueous buffers. Yet, they react when attached to biomolecules, e.g., due to protein binding, forming strong covalent bonds. This mechanism makes them promising in designing irreversible protein inhibitors and protein labeling probes.
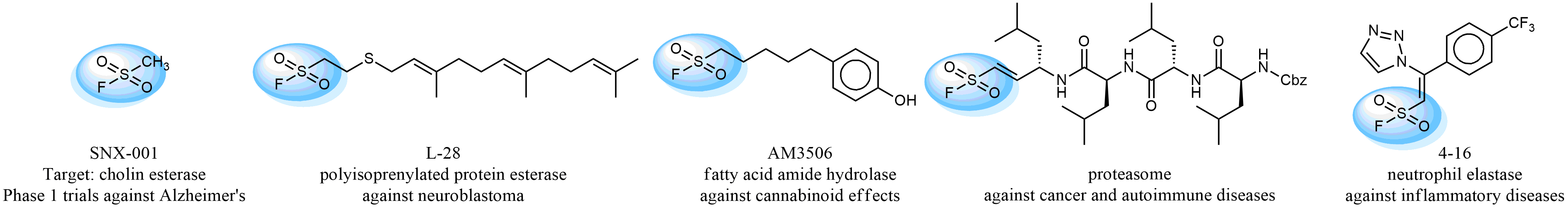
Case studies
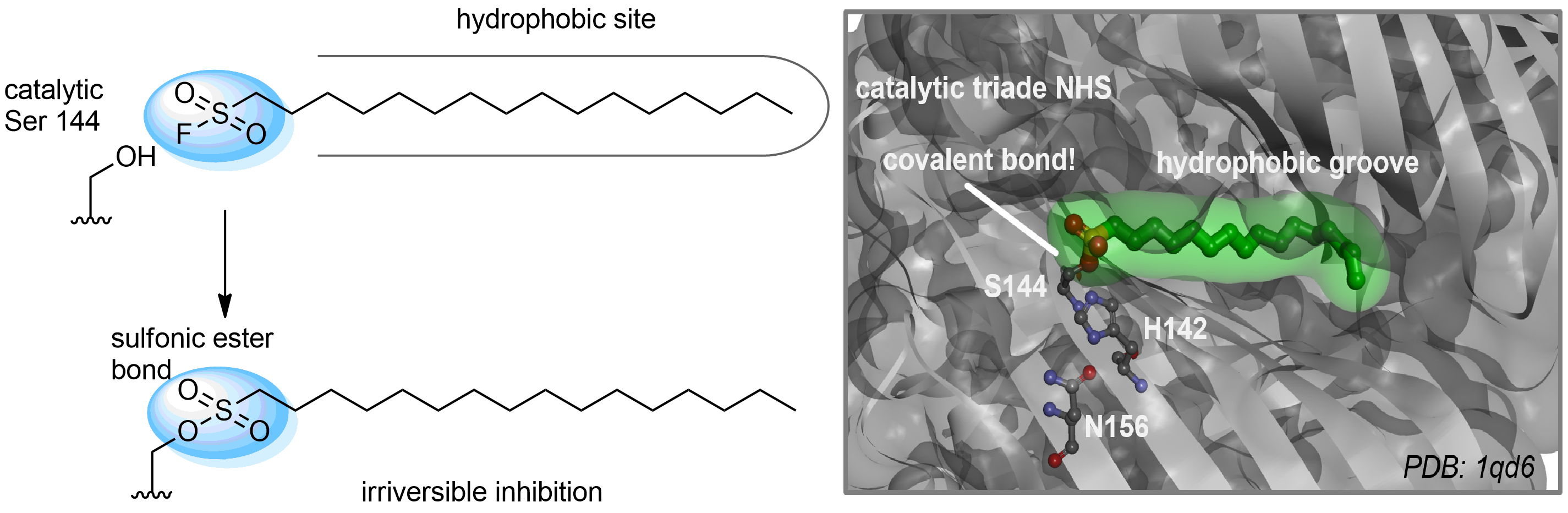
Hexadecanesulfonyl fluoride (AM 374) binds to the hydrophobic groove of the inner interface formed by homodimer of outer membrane phospholipase A. Subsequent formation of a sulfonic ester with the catalytic serine-144 leads to irreversible inhibition.
Download SD files
Download PDF file
We offer
More than 50 aliphatic sulfonyl fluorides from stock on a 5-10 g scale.