The ultimate selection of Lys-specific binders
1 600 compounds
Until recently, covalent binders targeting Lysine residue attracted less attention as compared to those modifying Cysteine or Serine. Being among the essential amino acids, Lysine can be found both at the surface and in active sites of many enzymes (e.g. kinases, viral polymerases and integrases, aldolases, DOPA decarboxylase, P-glycoprotein), and it can also participate in catalytic reactions. Moreover, Lys residue is often involved as a key player in many important signaling and metabolic processes. An example can be protein ubiquitination, which mainly occurs through lysine residues on substrate proteins or itself. Selective modification of Lys residue becomes more and more attractive for the investigation of many signaling processes in live cells and, potentially, can play a key role in the discovery of next-generation drugs.
Lysine focused Library is plated at 20 mM concentration in DMSO and is available for fast supply in any custom format. Compounds pooling based on delta MW can be provided as a formatting option. Please request the list of expected molecular weight shifts for MS-based screenings.
Typical Formats
Lysine-Focused Covalent Library is available for supply in various pre-plated formats, including the following most popular ones:
Catalog No.
LYS-1600-0-Z-10
Compounds
1 600
2 plates
Amount
≤ 300 nL of 10 mM of DMSO solutions
Plates and formats
1536-well Echo LDV microplates, first and last four columns empty, 1280 compounds per plate
Price
Catalog No.
LYS-1600-10-Y-10
Compounds
1 600
5 plates
Amount
10 µL of 10 mM DMSO solutions
Plates and formats
384-well, Echo Qualified LDV microplates #001-12782 (LP-0200), first and last two columns empty, 320 compounds per plate
Price
Catalog No.
LYS-1600-50-Y-10
Compounds
1 600
5 plates
Amount
50 μL of 10 mM DMSO solutions
Plates and formats
384-well, Greiner Bio-One plates #781280, first and last two columns empty, 320 compounds per plate
Price
Catalog No.
Library & follow-up package
Plates and formats
LYS-1600-10-Y-10 screening library 1 600 cmpds, hit resupply, analogs from 4.7M+ stock and synthesis from REAL Space
Price
*We will be happy to provide our library in any other most convenient for your project format. Please select among the following our standard microplates: Greiner Bio-One 781270, 784201, 781280, 651201 or Echo Qualified 001-12782 (LP-0200), 001-14555 (PP-0200), 001-6969 (LP-0400), C52621 or send your preferred labware. Compounds pooling can be provided upon request.
Download SD file
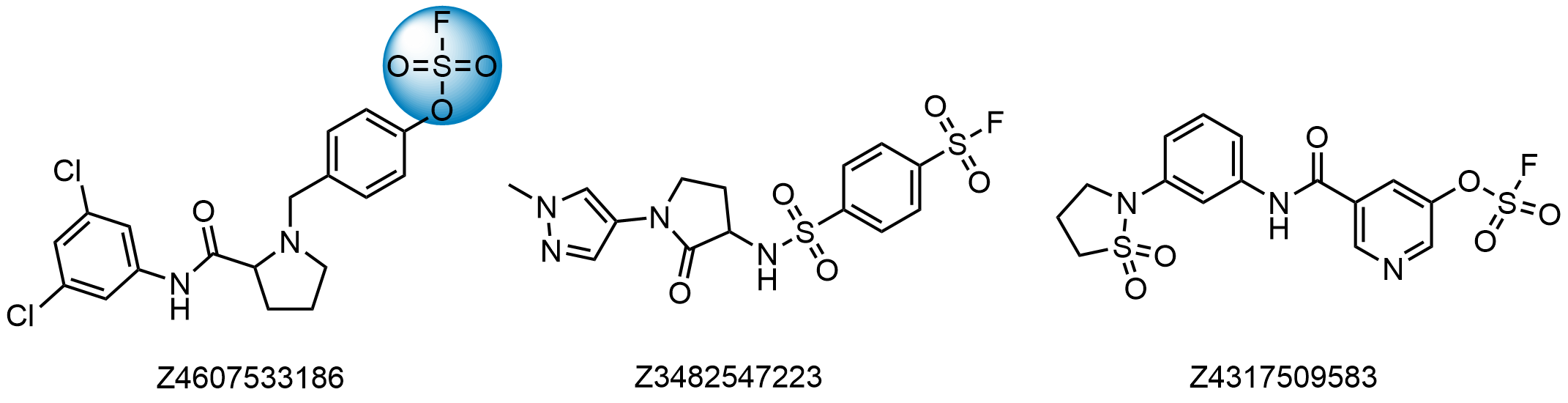
Examples of compounds in the library
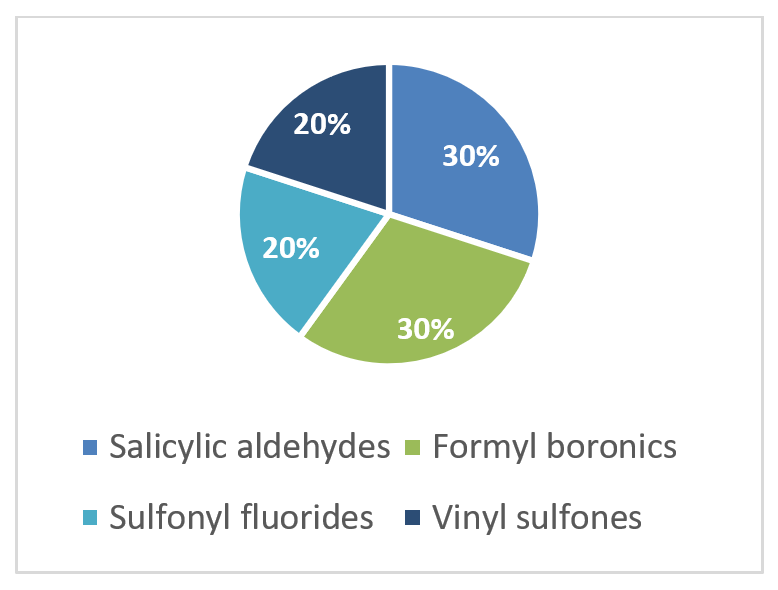
To address the increased interest and need for lysine-specific covalent binders, we have synthesized several libraries bearing warheads described as selective/preferred toward Lysine. Our renewed library contains only specially synthesized compounds with Lys-specific covalent warheads. The following types of covalent binders were used for the library construction:
Salicylic aldehydes
- Lys-specific, reversible
- The o-hydroxy group stabilizes covalent adducts
- Low reactivity
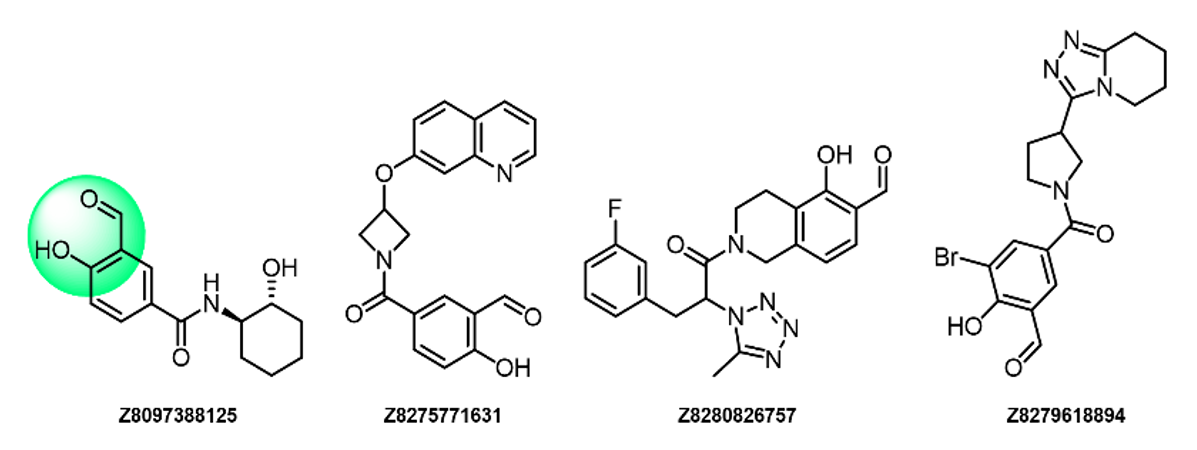
Vinyl Sulfonamides
- Irreversible binders
- Showed some selectivity toward Lys over Cys
- Highly reactive
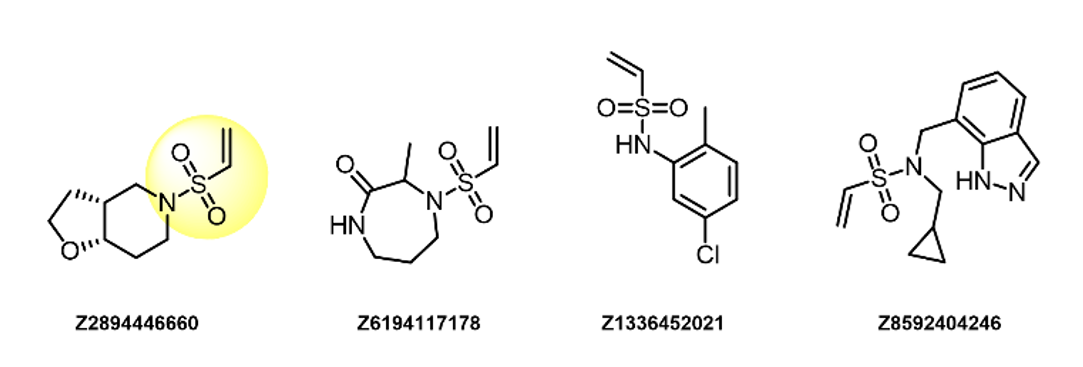
α-Formyl Boronic acids
- Reversible and Lys specific
- Boronic residue in α-position dramatically enhances the stability of resulting adducts
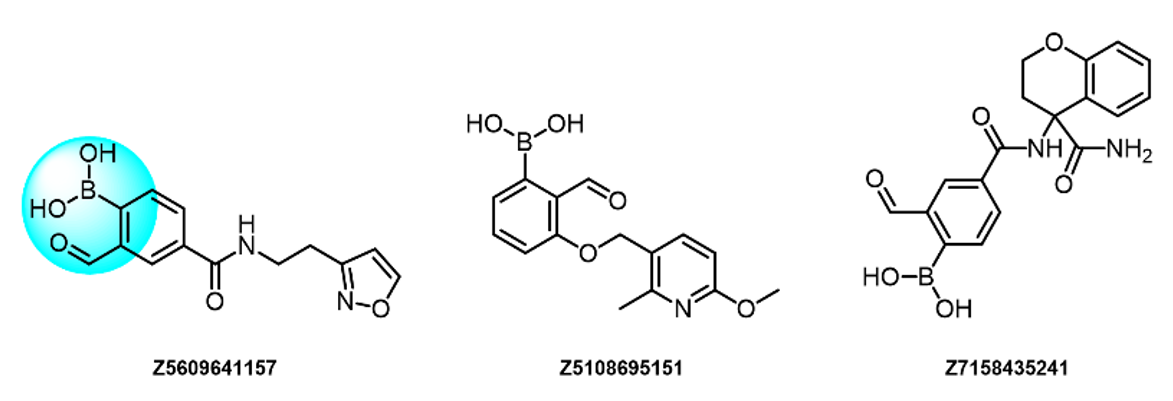
Sulfonyl Fluorides
- Irreversible binders for Lys, pH-dependent
- Reactivity depends on structure
- Compounds selected to display moderate reactivity
Distribution by covalent warhead
The library can be screened directly at Enamine in our biology laboratories, which offer a wide range of screening techniques including MS-based screening, kinetics, and intrinsic reactivity measurements. In this case, we will be happy to offer you a discount on library costs and follow-up services depending on the collaboration scope.



