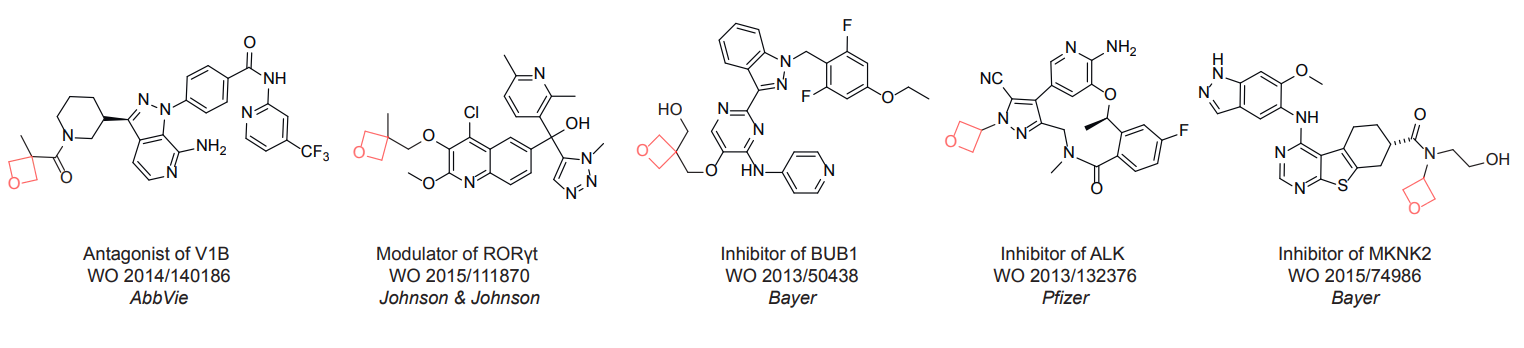
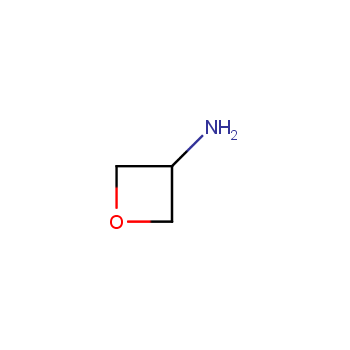
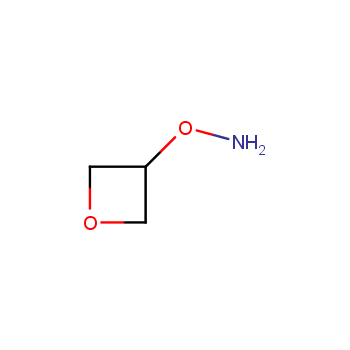
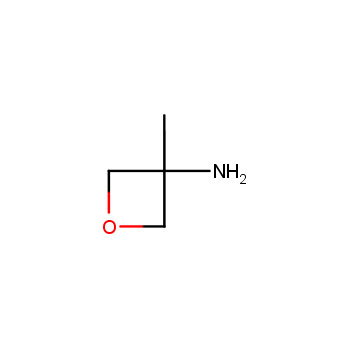
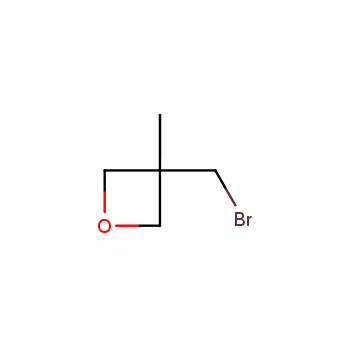
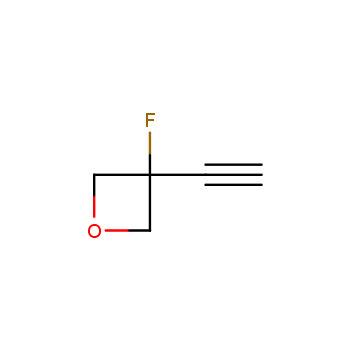
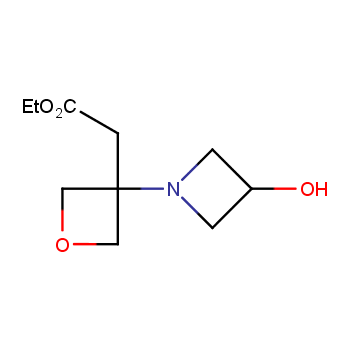
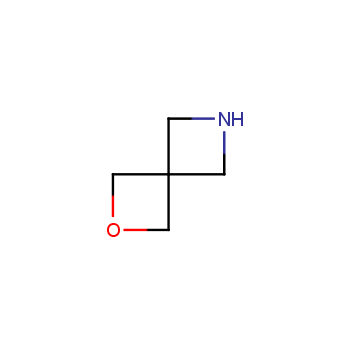
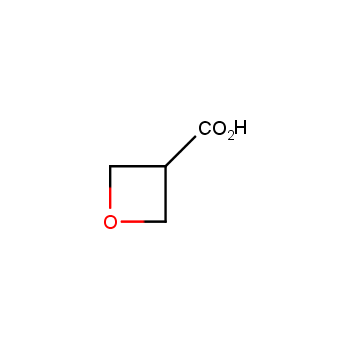
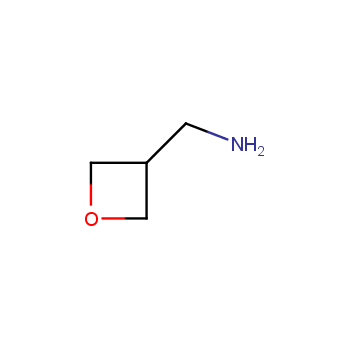
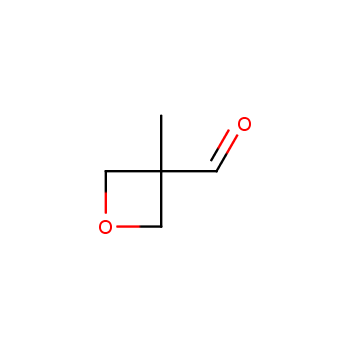
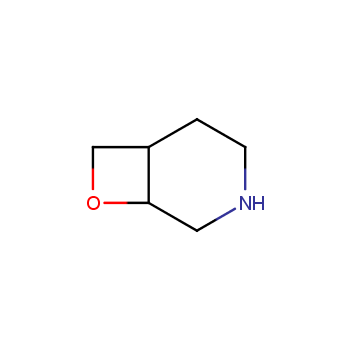
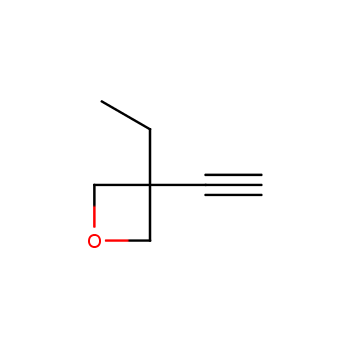
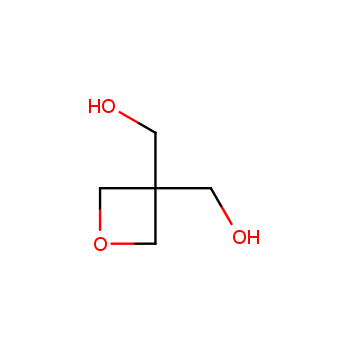
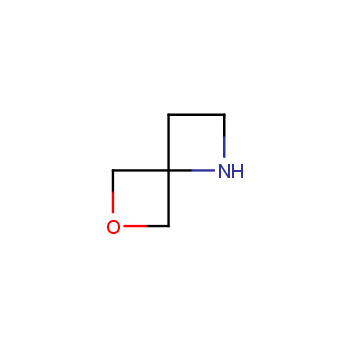
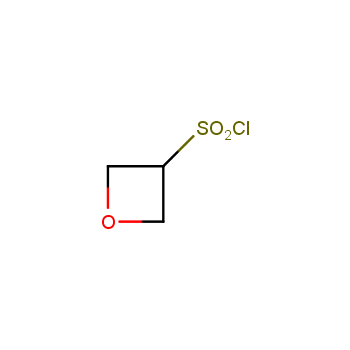
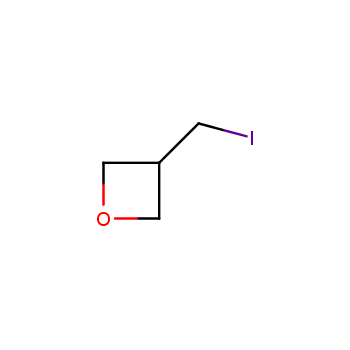
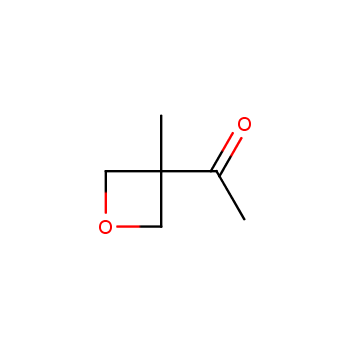
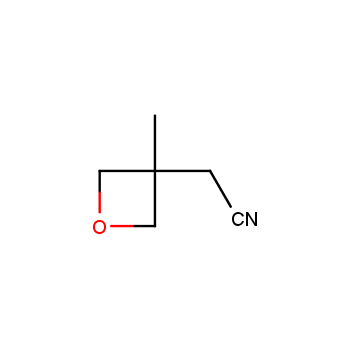
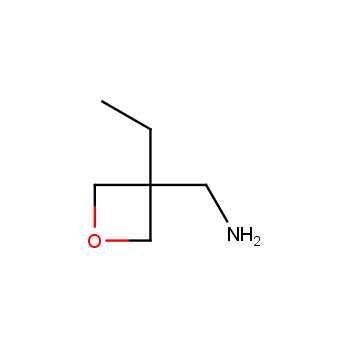
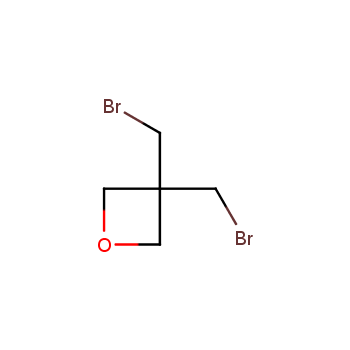
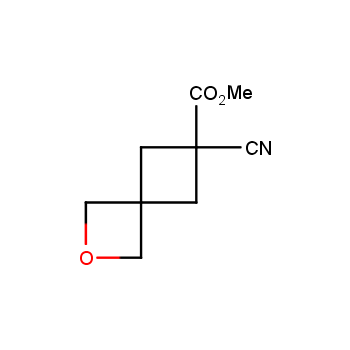
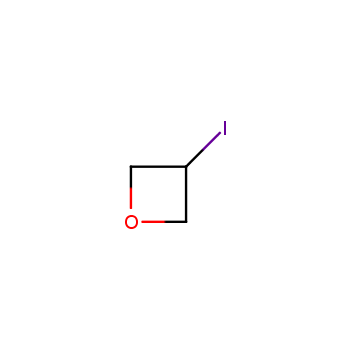
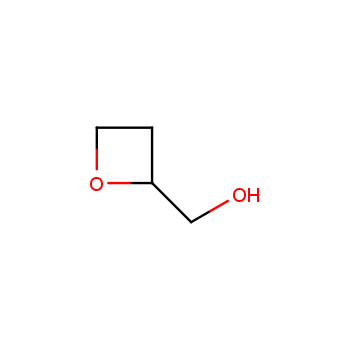
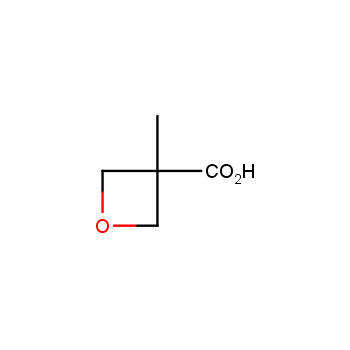
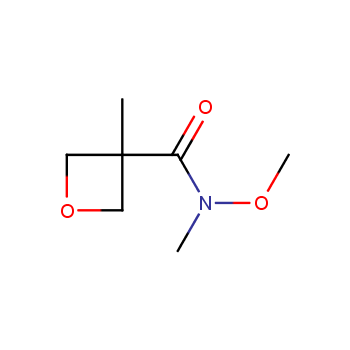
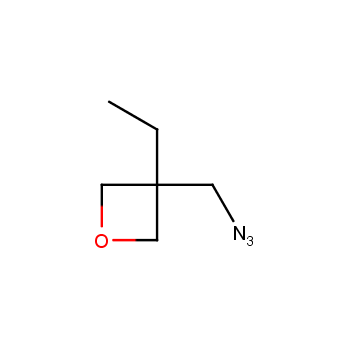
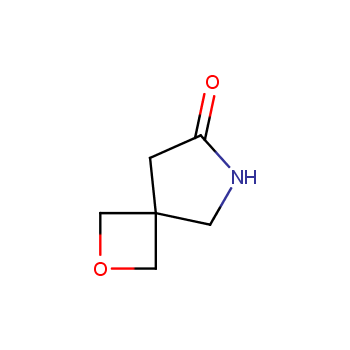
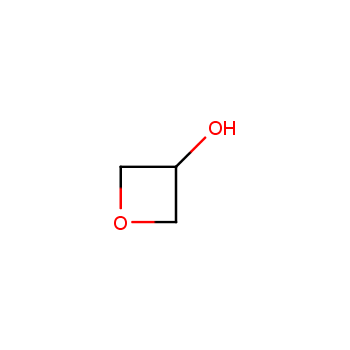
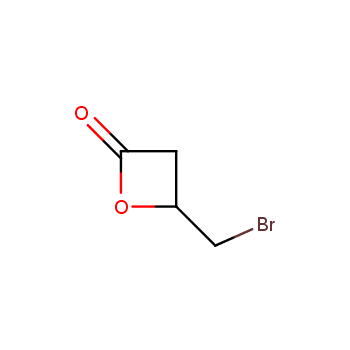
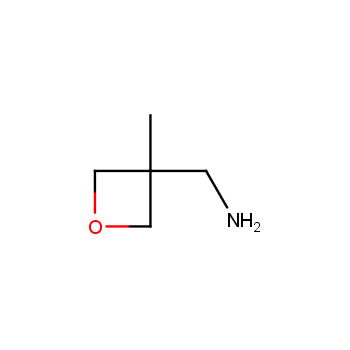
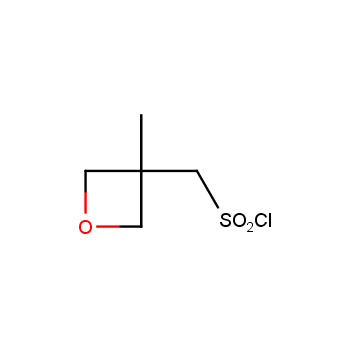
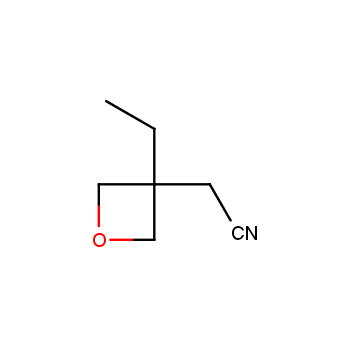
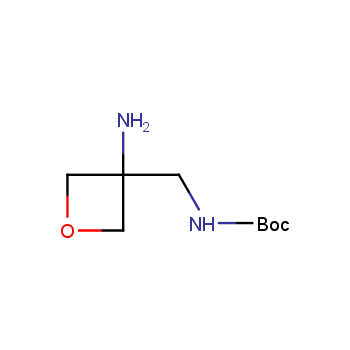
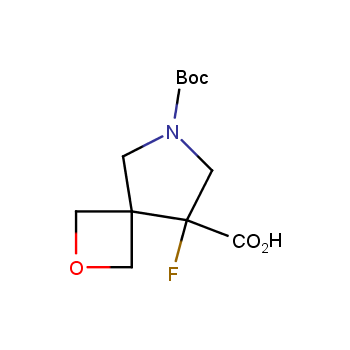
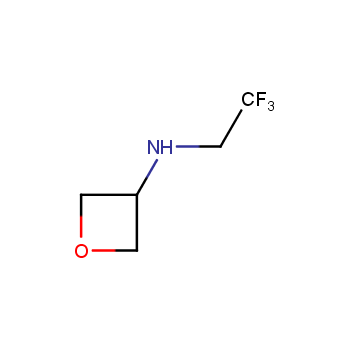
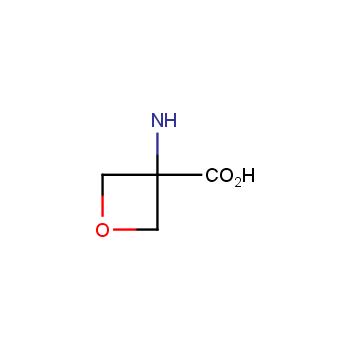
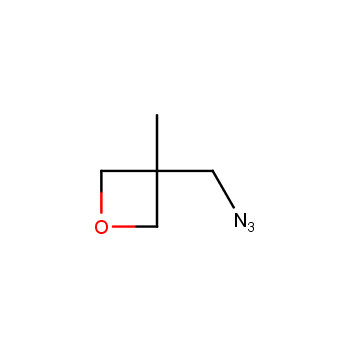
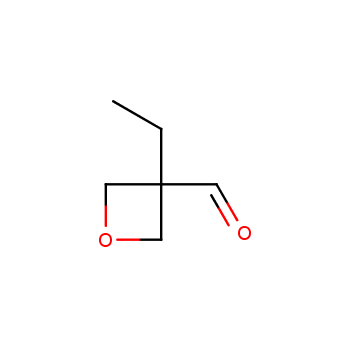
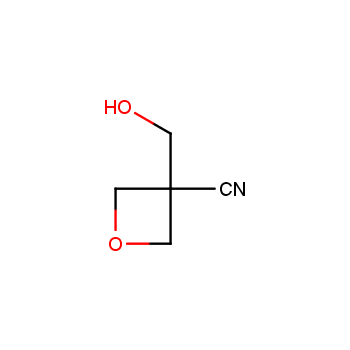
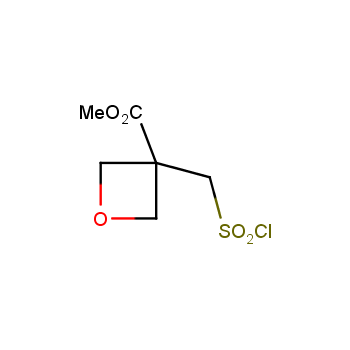
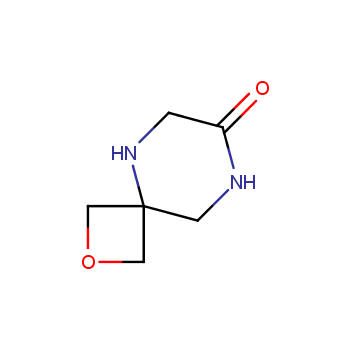
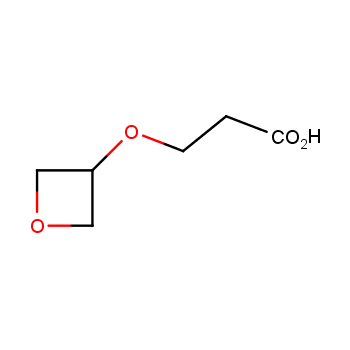
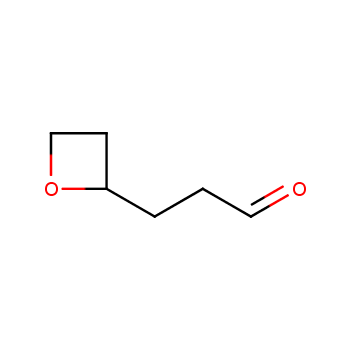
For more than 130 years since the first preparation by Reboul, oxetanes have largely remained neglected in medicinal chemistry. The unit of oxetane can trigger profound changes in aqueous solubility, lipophilicity, metabolic stability, and conformational preference when replacing the commonly employed functionalities such as gem-dimethyl or carbonyl groups. Of particular interest are the oxetanes substituted at the 3-position, since they remain none-chiral. At the moment, oxetane-containing building blocks flourish in medicinal chemistry and drug discovery.
Properties
- high chemical stability;
- high aqueous solubility;
- low lipophilicity;
- high metabolic stability;
- hydrogen-bond acceptor ability.
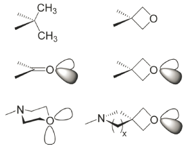
Application
- less lipophilic and more metabolically stable than a gem-dimethyl group;
- replacement for a metabolically and chemically labile carbonyl group;
- metabolically-robust analogue of morpholine.
Our offer:
>200 oxetane-containing building blocks on gram scale in stock.