300 Thousand compounds in stock
Original and unique
Make-on-demand
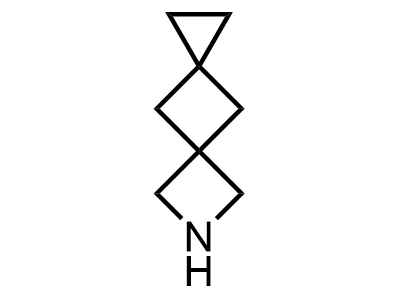
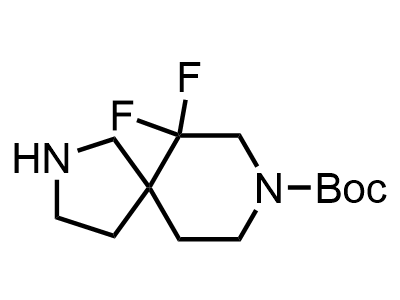
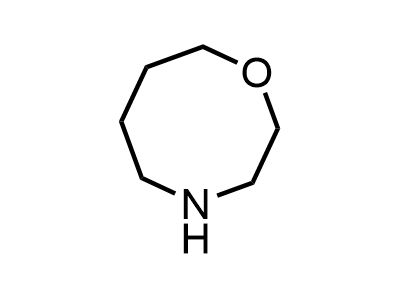
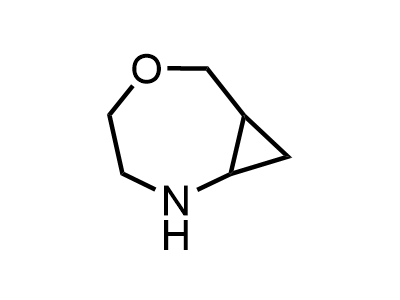
Building Blocks
1B novel building blocks
Reliable supply
Over 650 highly skillful chemists
Unique synthesis technologies
48B Billion
REAL compounds and
Custom Library Synthesis
On site access to all Enamine stock BB’s
Highly flexible arrangements
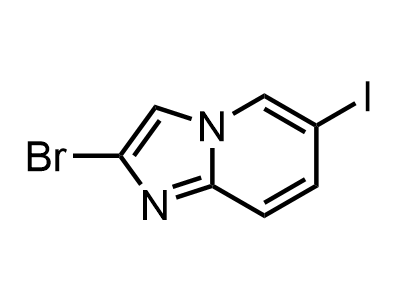
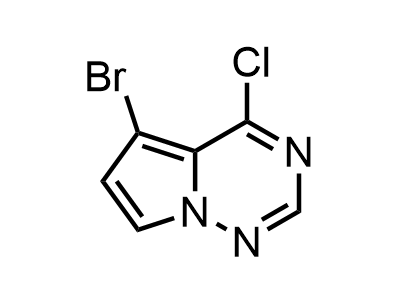
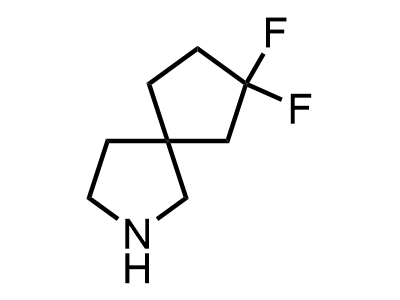
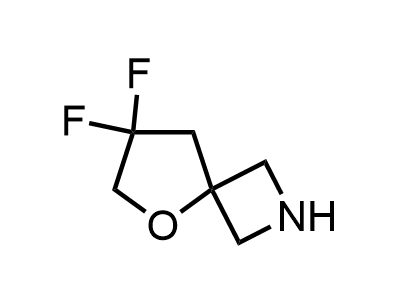
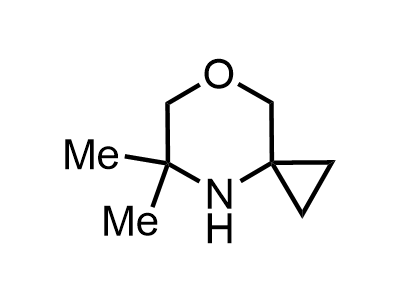
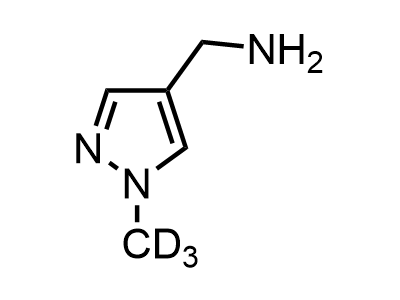
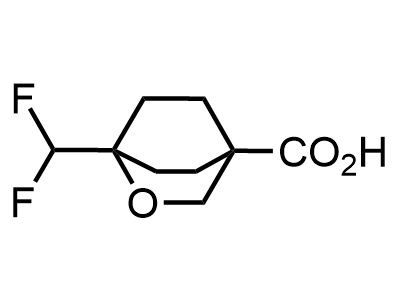
2 000 new building blocks are synthesized monthly. Here is an important update to our MedChem Highlights from March 2024
Recent News
11 April 2024
Press Release
Cambridge, UK and Kyiv, Ukraine, 11 April 2024: Metrion Biosciences Limited (“Metrion”), the specialist ion channel and cardiac safety screening contract research organisation (CRO) and drug discovery company, and Enamine Ltd (“Enamine”), the global leader in supplying small molecules and early drug discovery services, announced that Metrion has enhanced its High Throughput Screening (HTS) services with the addition of access to Enamine’s compound libraries.
27 March 2024
Press Release
March, 2024, Kyiv, Ukraine. Enamine Ltd, the global leader in supplying small molecules and early drug discovery services, announces the expansion of its library synthesis capabilities with a focus on Enamine REAL compounds to further support the growing demands of agricultural and pharmaceutical companies, research institutes, and drug discovery centers.
01 March 2024
News
We are excited to announce a strategic collaboration between Enamine, the world's leading provider of chemical building blocks, compound libraries, and biology services, and Genez International, a prominent enterprise with 15 years of experience in cross-border supply management, biopharmaceutical research and development, semiconductor equipment, and high-definition digital imaging systems.
J. Heterocycl. Chem.
2019, 56 (5), 1605-1612
DOI:
10.1002/jhet.3541
Voskoboynik O.; Kolomoets O.; Nosulenko I.; Berest G.; Bilyi A.; Karpenko O.; Belenichev I.; Kovalenko S.
Previously unknown 3′‐R1‐5‐R2‐spiro[indoline‐3,6′‐[1,2,4]triazino[2,3‐c]quinazoline]‐2,2′‐(7′H)‐diones and their N‐substituted analogues were obtained via reaction of 6‐R1‐3‐(2‐aminophenyl)‐1,2,4‐triazin‐5‐ones with isatin and its substituted derivatives. It was shown that alkylation of 3′‐R1‐5‐R2‐spiro[indoline‐3,6′‐[1,2,4]triazino[2,3‐c]quinazolin]‐2,2′‐(7′H)‐diones by N‐R3‐chloroacetamides or chloroacetonitrile in the presence of а base proceeds by N‐1 atom of isatin fragment. The spectral properties (1H and 13C NMR spectra) of synthesized compounds were studied, and features of spectral patterns were discussed. The high‐effective anticonvulsant and radical scavenging agents among 3′‐R1‐5‐R2‐spiro[indoline‐3,6′‐[1,2,4]triazino[2,3‐c]quinazolin]‐2,2′(7′H)‐diones and their N‐substituted derivatives were detected. It was shown that compounds 2.2, 2.8, and 3.1 exceed or compete the activity of the most widely used in modern neurology drug—lamotrigine on the pentylenetetrazole‐induced seizures model. The aforementioned fact may be considered as a reason for further profound study of synthesized compounds using other pathology models.
![Synthesis, Modification, and Anticonvulsant Activity of 3′-R 1 -Spiro[indoline-3,6′-[1,2,4]triazino[2,3-c]quinazolin]-2,2′(7′H)-diones Derivatives](/images/publications/2019/jhet.3541.PNG)
![Synthesis, Modification, and Anticonvulsant Activity of 3′-R 1 -Spiro[indoline-3,6′-[1,2,4]triazino[2,3-c]quinazolin]-2,2′(7′H)-diones Derivatives](/images/publications/2019/jhet.3541.PNG)